What occurs when an atom loses electrons?
Oxidation
What is silver attracted to during its electroplating process?
To the cathode
What occurs when a atom gains electrons?
Reduction
What are reactions that occur in each half-cell?
Half-reactions
What is a half cell?
A section of a voltaic cell where either oxidation or reduction occurs
What is happening to an oxidizing agent in a redox reaction?
It is being reduced
What oxidation number does free elements have?
0
What is the Hall-Herout process?
The electrolysis process used to produce aluminum in industry. It converts bauxite into aluminum.
What is a salt bridge?
A tube of electrolytic gel that connects the two half-cells of a voltaic cell that allows the flow of ions but prevents the mixing of the solutions
What is electrolysis?
The process of forcing an otherwise nonspontaneous redox reaction to occur with the aid of an electrical current in an electrochemical cell.
Indicate if the following reaction is a redox reaction or not. If it is a redox reaction, state the oxidizing and reducing agent.
3Cl2 + 6NaOH → 5NaCl + NaClO3 + 3H2O
It is a redox reaction
Oxidizing agent: Cl2
Reducing agent: NaOH
What is the difference between electrolytic cells and voltaic cells?
Electrolytic cells - use electrical energy to force a nonspontaneous chemical reaction to occur
Voltaic cell - uses a spontaneous redox reaction to produce electrical charges
Indicate if the following reaction is a redox reaction or not. If it is a redox reaction, state the oxidizing and reducing agent.
Na2S + 2HCl → 2NaCl + H2S
Not a redox reaction
On a voltaic cell, which part is positive and which part is negative?
Anode is negative
Cathode is positive
Write the half reactions and full redox reaction for the oxidation of iron into iron (III) and the reduction of vanadium (II) ions.
Half: Fe → Fe3+ + 3e-
V2+ + 2e- → V
Redox: 2Fe2+ + 3V2+ → 2Fe + 3V
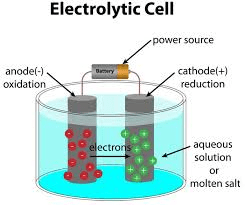
Draw an electrolytic cell with a cathode, anode, power source, and electrolyte solution. Indicate the charges of the cathode and anode
Draw a voltaic cell with a cathode, anode, voltmeter, and salt bridge. Indicate the charges of the cathode and anode.

Give four examples of voltaic cells.
Rechargeable batteries
Lead-acid storage batteries
Lithium batteries
Fuel cells