This is what is being dissolved
Solute
The three ways to increase the solubility of a solid:
agitation
increase temperature
increase surface area
What is the % concentration of a solution if you put 15.0 g of NaCl in 100.0 g of a solution?
15%
What is the molarity if 0.235 moles of NaCl are dissolved in 3.5 L of solution?
0.067 M
Colligative Properties are dependent on _______
the identity of the solute
A type of solution in which you can add more solute
unsaturated
list the two ways to increase the solubility of a gas
decrease temperature
increase pressure
What is the % concentration of a solution if you put 15.0 g of NaCl in 100.0 g of WATER?
13.04%
How many moles of silver nitrate would you need to make a 5M solution of silver nitrate with a volume of .5L?
2.5 moles
Oil is Nonpolar
Water is Polar
So Br2 will dissolve in _________
oil
liquid dissolving in another liquid is called ______
miscible
Identify the two curves that behave like a gas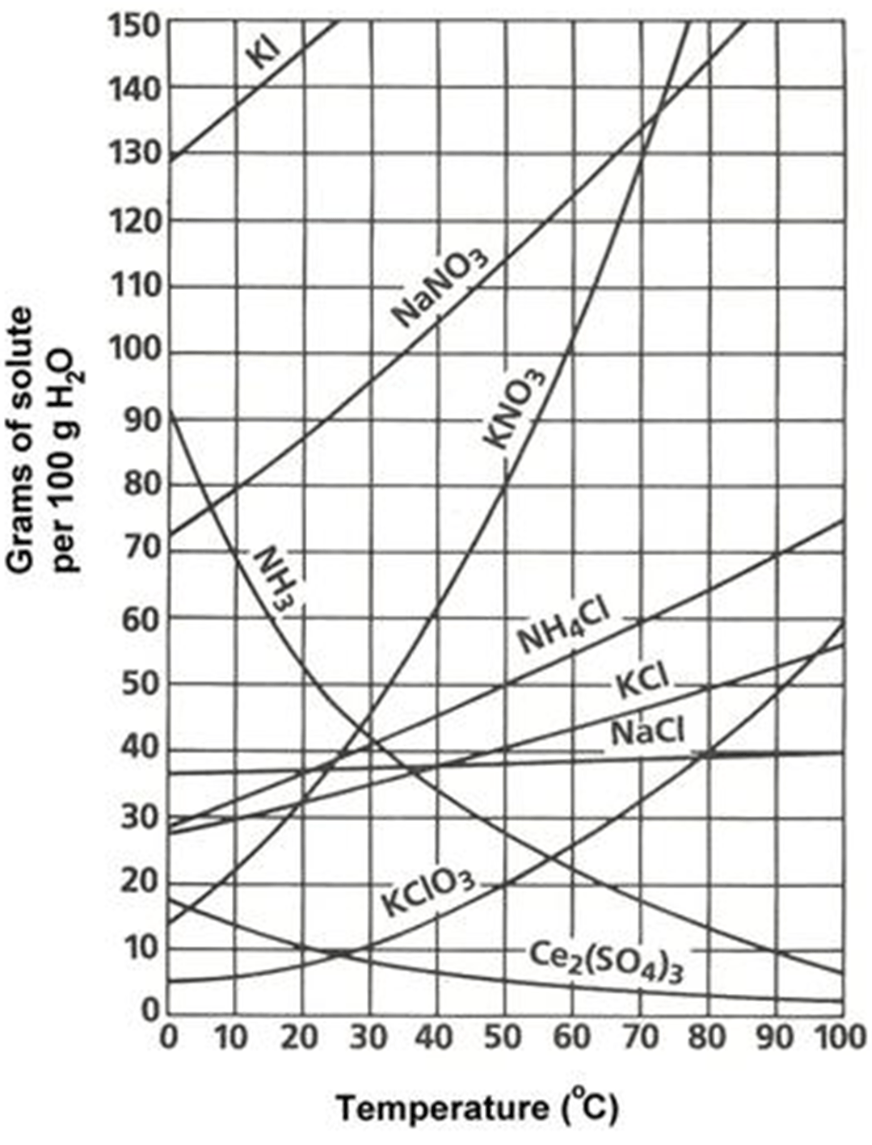
NH3
Ce2(SO4)3
How many grams of NaH2CO3 are in 100.0 g of a 20.0% solution?
20 g
What volume would a 1.15 M solution of potassium nitrate need to make 75.83 grams of KNO3?
.65 L
Polar or NonPolar:
C12H78
Nonpolar
when ionic compounds dissolve in water it ___________ then it _______________
dissociates; solvates/solvation
A solution containing KNO3 was increased from 50C to 70C, how much more can I add to the solution?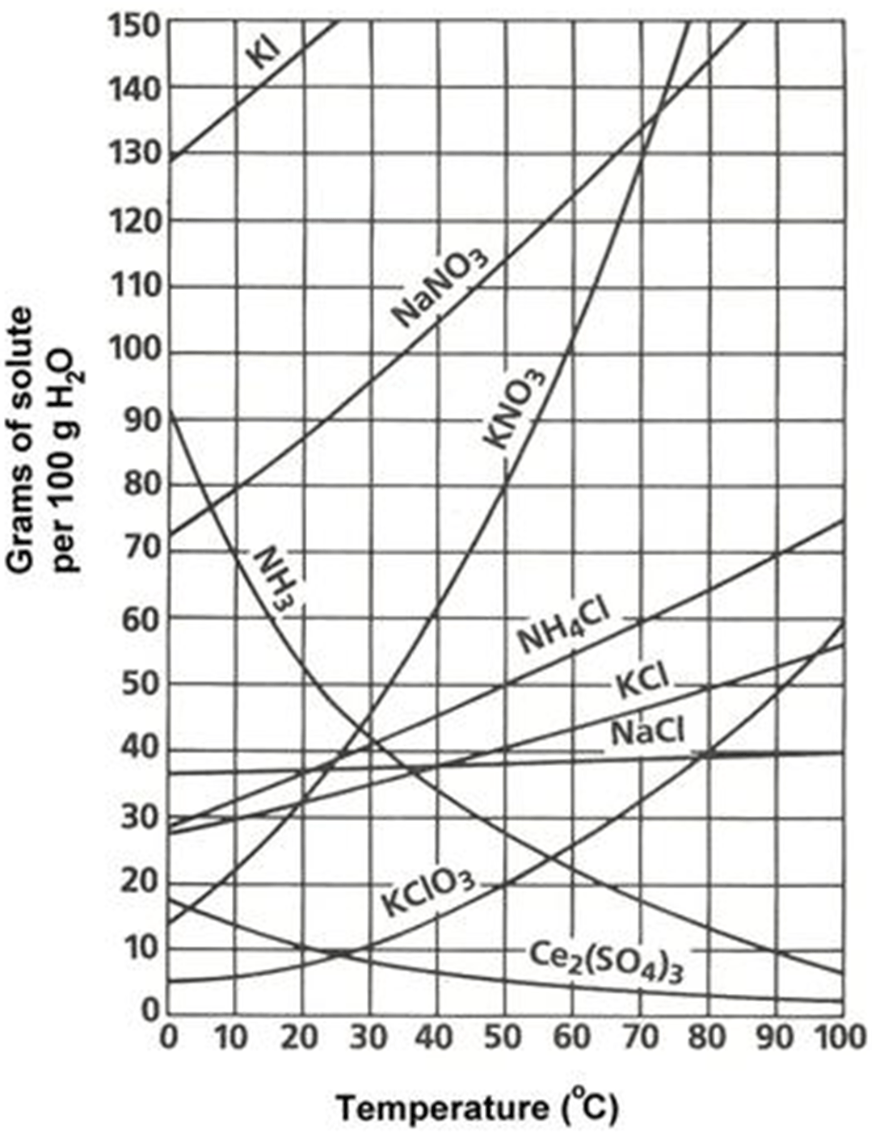
50 g
If we dissolve 25.0 g KCl in a solution and it has a 20.0% concentration, what is the mass of the solution?
125 g
What is the molarity of an aqueous solution that contains 14.2 g NaCl dissolved in 23 mL of solution?
10.6 M
Polar or Nonpolar
HCl
Polar
If I were to add more sugar to a sugar solution and crystals start to form, this is described as a _____________ solution
+100 if you can tell me if it is stable or unstable
Supersaturated
Unstable
How many grams of KCl could dissolve in 135 g of water at 40 °C?

About 54 grams
A KNO3 solution is 6.25% and contains 20.0 g KNO3. What is the mass of the solution? What is the mass of the solvent?
320 g solution
300 g solvent
How many grams would I need to make a 623 mL solution of 0.7 M CuSO4?
69.61 g
Why do we throw salt on the sidewalks before it snows?
Decrease the freezing point so the sidewalks do not ice over
Which of the following would have the greatest effect on colligative properties?
MgSO4
NaCl
Al2(CO3)3
FeCl3
Na2SO4
Al2(CO3)3