Give an example of an ionic compound.
Give an example of a covalently bonded molecule.
Any sensible suggestion
What charge does a potassium atom have once it is bonded via metallic bonding to other potassium atoms?
+1
Name two giant carbon structures.
Diamond/graphite/buckyball/graphene etc
Name two types of smart material.
Shape memory polymer, shape memory alloys, photochromic materials, thermochromic materials, hydrogels
What do we call an atom that has lost or gained an electron and now has either a positive or negative charge?
An ion
Covalent bonding happens between ...................... (what type of elements?)
Covalent bonding happens between two non metals
Metal atoms (or positively charged ions) are held together by a ....................................
Metal atoms (or positively charged ions) are held together by a sea of electrons.
What shape is buckminsterfullerene?
Why would some people be against the use of silver nanoparticles?
Easily penetrate the body, could cause side effects, easily released into the environment, more research is needed to fully understand them.
List three properties of ionic compounds.
-Made of crystals (as they form lattices)
-High melting points/high boiling points
-Often soluble in water
-Conduct electricity when molten
Why does methane (CH4), a simple covalent structure have a much lower melting point than a giant covalent structure like diamond?
As they are only small molecules, the forces between the molecules is weak and can be overcome easily. With giant covalent structures, you need to break all the covalent bonds before it turns to a liquid.
Draw a diagram to show metallic bonding.
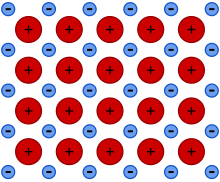
Explain why graphite can conduct electricity.
Each carbon atom is bonded to three others, so each atom has a free electron, resulting in a sea of electrons that can carry electrical charge.
How small is a nanometre?
One billionth of a metre
Why would an ionic compound with magnesium have a higher melting point than a compound with sodium? (Hint: think of group number, and what this means for the ions formed)
Magnesium is in group 2, whereas sodium is in group 1. When magnesium forms an ionic compound it becomes a 2+ ion, and so the attraction to the negative ion in the compound is much stronger, and hence the melting point will be much higher.
Draw the covalent bonding as a dot-cross diagram for NH3

Explain why metals can conduct electricity.
Metals conduct electricity because they have “free electrons.” Unlike most other forms of matter, metallic bonding is unique because the electrons are not bound to a particular atom. This allows the delocalized electrons to flow in response to a potential difference.
List two uses of nanotubes.
Drug delivery systems, as catalysts, to reinforce materials, as lubricants for machinery
Explain what thermochromic and photochromic smart materials do.
Thermochromic: materials that change colour as a result of temperature
Photochromic: materials that change colour as a result of light
Draw a dot-cross diagram to show the ionic bond in the compound Li2O.

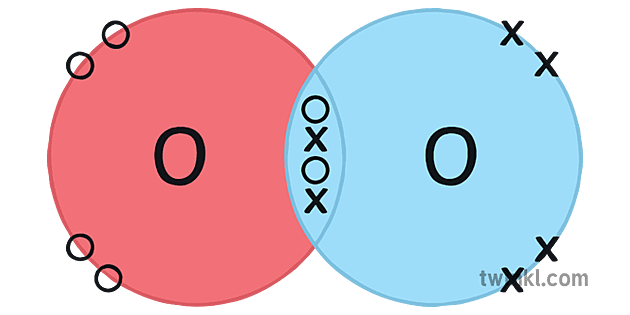
Draw the covalent bonding in O2 as a dot-cross diagram.
List six properties of metals.
-Shiny
-Malleable
-Ductile
-Hard
-Dense
-Can conduct electricity
-High melting and boiling points
Draw the giant covalent structure of graphite.
Name two compounds that are used as nanomaterials.
Silver, zinc oxide, titanium oxide