The number of grams of Ca(OH)2 contained in 0.500 L of a 0.450 M solution.
What is 16.7 g of Ca(OH)2?

Amount of K2Cr2O7 that will precipitate out of a saturated solution at 80oC, when cooled to 30oC.
What is 45 g K2Cr2O7
This TV character said, “Live long and prosper”
What is the molality of a solution that contains 48 grams of sodium chloride and 250 mL of water?


30.0 g NaCl is added to 50.0 g H2O at 50oC, determine if the resulting solution will be saturated, unsaturated or superaturated
What is saturated? (@ 50oC 100g of water can hold about 35g NaCl, so with 50g of water you can dissolve a maximum of about 18g of NaCl)
Write the formula for the insoluble compound created when the following pairs of solutions mix. If both products are soluble say no reaction.
1. iron (II) carbonate and sodium hydroxide
2. ammonium chloride and strontium perchlorate
3. barium nitrate and potassium sulfate
1. Fe(OH)2
2. no reaction
3. BaSO4
The density of ethylene glycol (antifreeze, HOCH2CH2OH) is 1.09 g/mL. How many mL of ethylene glycol should be mixed with 375 mL of water to make a 7.50% by mass mixture? (density of water is 1.00 g/mL)
x/(x+375) * 100 = 7.50
x/(x+375) = 0.0750
x = 0.0750x + 28.125
0.925x = 28.125
x = 30.4 g antifreeze
27.9 mL are required
The precipitate that will form when solutions of Sr(NO3)2 and K2SO4 are mixed
What is SrSO4?
What is the molarity of a solution that contains 10.0 grams of silver nitrate that has been dissolved in 750 mL of water? What is the percent by mass of silver nitrate? What is the mole fraction of the solute in the solution?
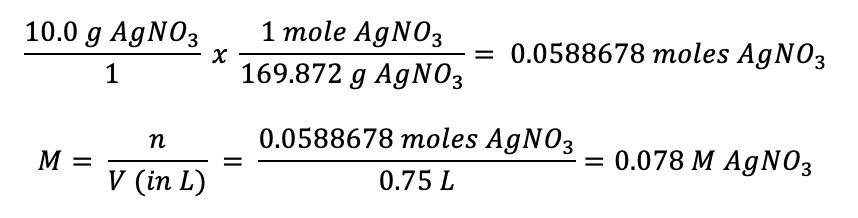


Write the chemical equation for the following reaction including states of matter: ammonium sulfate + aluminum chloride ->
(NH4)2SO4(aq) + AlCl3(aq) --> No reaction
Write the net ionic equation for the following reaction including states of matter: sodium carbonate + iron (II) chloride ->
CO32-(aq) + Fe2+(aq) -> FeCO3(s)
The average human body contains this many pints of blood.
What is nine?