The attractive forces between polar molecules.
What are intermolecular forces?
The electrons in the highest energy level, furthest from the nucleus.
What are valence electrons?
The rule dictating the number of bonds Carbon, Hydrogen, Nitrogen, and Oxygen tend to make in covalent compounds.
What is HONC 1234?
The molecular shape of water H2O.
What is bent?
A difference in electronegativity of 0.5 or greater makes a bond ______.
What is polar?
The rule that states that atoms will share electrons until their valence shell is full (usually 8 electrons)
What is the octet rule?
The Lewis Dot structure for H2O (including all lone pairs).
The smell of a molecule that contains nitrogen.
What is a fishy smell?
The reason some molecules with two bonds are bent and others are linear.
What are lone pairs?
The most electronegative atom on the periodic table.
What is fluorine?
A chemical property that describes the tendency of an atom to attract shared electrons
What is electronegativity?
The Lewis Dot Structure of CO2 (including ALL electrons).
::O=C=O::
The chemical formula for butyric acid:
What is C4H8O2?
The shape of an atom with 4 bonds, each 109.5 degrees apart.
This type of molecule is not attracted to a charged wand and will spread out on wax paper.
What is a non-polar molecule?
A pair of equal and oppositely charge polls separated by distance.
What is a dipole?
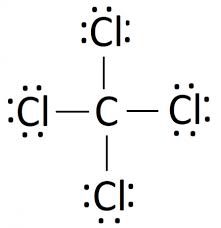
The Lewis Dot Structure for CCl4 (including all lone pairs)
The structural formula for C3H9N.
Many right answers, so long as HONC 1234 and correct number of atoms.
The shape of an atom with 3 electron domains and 0 lone pairs.
Trigonal planar
This type of molecule tends to have a smell.
What is a small, polar molecule?
A molecule that has the same chemical formula as another molecule, but a different 3D structure
What is an isomer?
The number of electron domains in NH3
What is 4 (3 bonds and 1 lone pair).
A structural formula for C4H8O.
Many potential answers, as long as they follow HONC 1234 and have the right number of atoms.
The molecular shape of H2CO.
What is trigonal planar?
The reason that CO2 is not polar.
Symmetrical/ Dipoles Cancel Out.