The type of bond found between atoms within a water molecule is.... Answer in two words
Polar Covalent
Which type of bond in water molecules rapidly break and reform?
Hydrogen Bonds
This is due to the partial negative and positive charges of the shared electrons
When does capillary action occur?
I. When cohesion is greater than adhesion
II. When adhesion is greater than cohesion
II. When adhesion is greater than cohesion
What is the difference between hydrophilic and hydrophobic molecules?
Hydrophilic: water-loving, polar, dissolves in other polar solvents
Hydrophobic: repels water, non-polar, dissolves in other non-polar solvents
Define Viscosity
Resistance to flow is related to the amount of energy required to change the shape of a liquid. Water has a low viscosity, but a greater viscosity than air.
State which element of water exerts a stronger pull on electrons
Oxygen
Oxygen is more electronegative than hydrogen, which means it attracts the shared electrons with more force
Which part of a water molecule has a partially negative charge and a partially positive charge
Oxygen side (negative pole)
Hydrogen side (positive pole)
Which of the following is NOT a real-life example of adhesion?
A. surface tension on the surface of a lake
B. Dew drops hanging off the tip of a blade of grass
C. water coating the surface of the alveoli
D. Water sticking to cellulose in cell walls
Choice A
Surface tension on the surface of the lake, describes the property of cohesion, the ability of water molecules to form hydrogen bonds with other water molecules. Choice A is correct.
Which of the following statement(s) about the role of water as a solvent is correct?
I. Salt can dissolve in water because it's hydrophilic
II. Lipids cannot dissolve in water because they are non-polar
III. Cellulose can dissolve in water, as it is able to form hydrogen bonds
I and II only
Why is the ringed seal able to float on the surface of water?
Low-density blubber increases the buoyant force
Which is the correctly annotated diagram of a molecule of water? 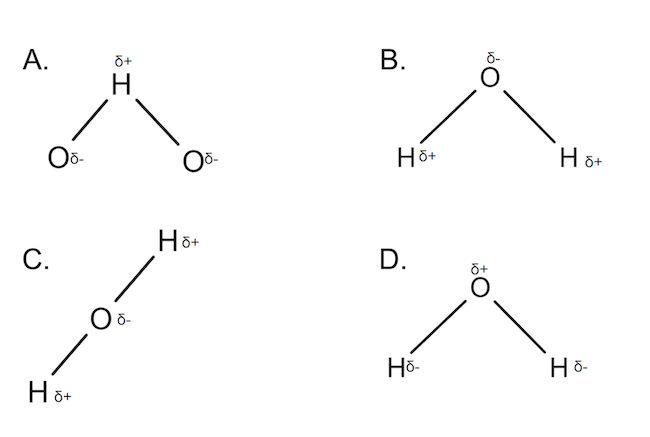
Choice B!
A water molecule consists of 2 hydrogens and one oxygen connected by single covalent bonds. There is a slight negative charge on the oxygen and a slight positive charge on the hydrogens due to unequal sharing of electrons
At what angle do the hydrogen atoms sit?
Why do hydrogen atoms repel each other?
104.5 degree angle because the pair of electrons repel each other. they are both positively charged
The diagram below shows a water column inside a xylem vessel:
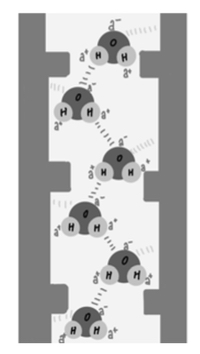
Which of the following best explains how the property illustrated contributes to plant survival?
I. Cohesion between molecules allows tension to be transmitted up the xylem
II. Cohesion enables water to evaporate more easily
III. Adhesion prevents the water column from breaking
Statements I and III
Statement I is true because cohesion refers to the attraction between water molecules due to hydrogen bonding. This cohesive property allows the water column to behave as a continuous stream, enabling tension created by transpiration at the leaves to be transmitted all the way down the xylem. This is central to the cohesion-tension theory of water transport.
Statement II is false because cohesion actually resists evaporation. The hydrogen bonds between water molecules make it harder for individual molecules to escape into the air. Evaporation is more closely influenced by temperature and humidity, not directly by cohesion.
Statement III is true because adhesion is the attraction between water molecules and the walls of the xylem vessel. This helps to counteract gravity and supports the column, reducing the chance of it breaking, especially under tension during transpiration. So this contributes to the integrity of the water column.
Why is water considered a universal solvent and why is this important for the origin of life?
I. it dissolves a wider variety of substances than any other liquid
II. It can dissolve both polar and non-polar molecules
III. It enables complex biochemical reactions by facilitating the interaction of reactants
I and III only
Water is known as the "universal solvent" because it can dissolve a wider range of substances, than any other liquid (Statement I). This property is due to water's polarity and ability to form hydrogen bonds with various molecules. Water’s ability to dissolve various substances means that it can bring reactants together in a solution, enabling the complex biochemical reactions that are necessary for life (Statement III). Choice C is correct.
Which term to describe a property of water is matched to its correct definition?
A.) Exerts buoyant forces: much energy is required to turn water into a vapour
B.) High specific heat capacity: Much energy is required to raise the temperature of 1kg water by 1 degree celsius
C.) High boiling point: Turns from ice into steam at 100 degrees celsius
D.) Low viscosity: Easily allows objects to float on the surface
Choice B!
Water has a high specific heat capacity, meaning it can absord or lose a lot of heat with only a small temperature change. This property helps maintain stable temperatures in organisms and aquatic environments.