Anything that has mass.
What is matter?
The number of protons or electrons.
What is the atomic number?
Negatively charged subatomic particle.
What is an electron?
The smallest unit of an element that retains the chemical properties of that element?
What is an atom?
Dmitri Mendeleev
Who primarily organized periodic table?
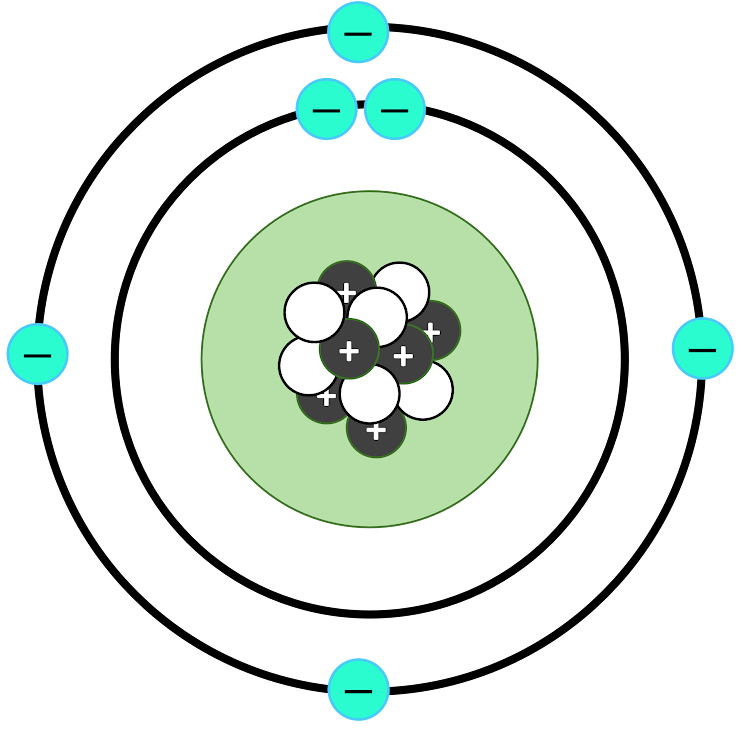
What is a Bohr model?
The number of protons and neutrons.
What is the atomic mass?
It is one (1).
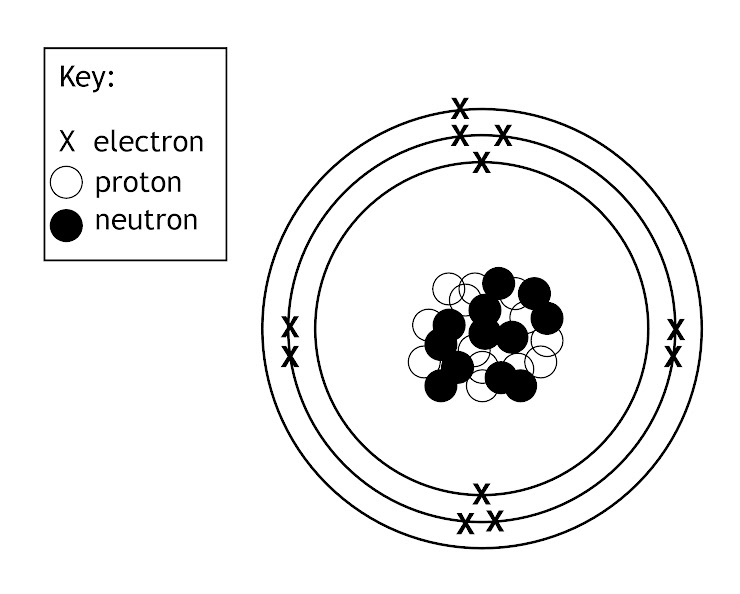
How many valence electrons are present?
Electrons orbit the nucleus in fixed energy levels.
What is a Bohr Model?
It is where hydrogen is located on the periodic table.
Which element is located in Group 1 (alkali metals) of the periodic table?
That is where protons and neutrons are found.
What is the nucleus?
It looks like this.
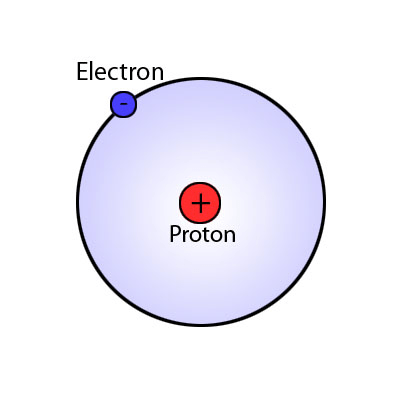
What is a Bohr model or the Bohr model of hydrogen?
29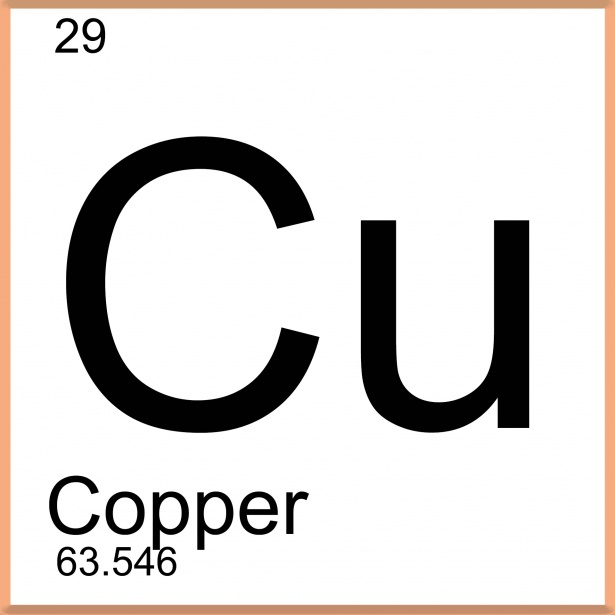
What are the number of protons or electrons?
The number of Valence electrons.
What the group of the Periodic Table can tell us?
They border the "zigzag" or "stair step" line.
What are metalloids?
Positively charged subatomic particles
What are protons?
A full octet.
What is a full valence shell?
35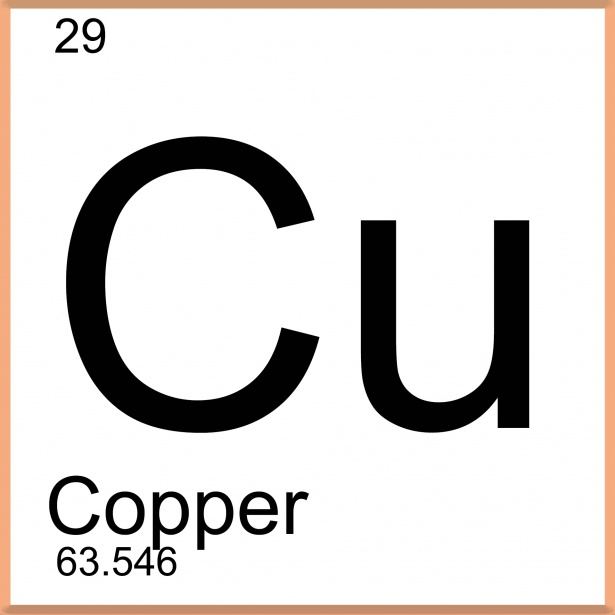
What are the number of neutrons in copper?
They are generally shiny and can conduct electricity.
What are the characteristic of metals?
An element that conducts electricity only under certain conditions.
What is a semiconductor?
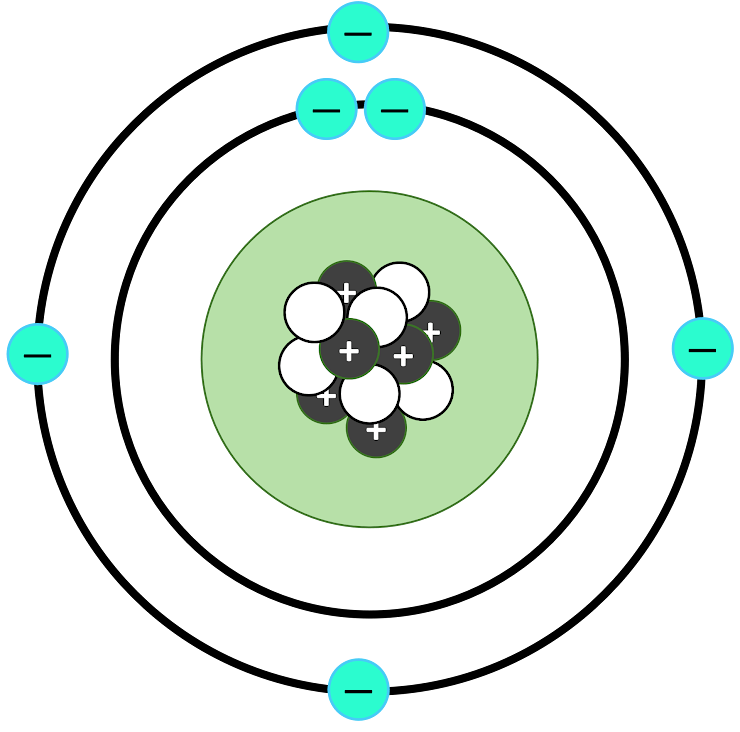 This is the element's Bohr Model
This is the element's Bohr Model
What is carbon?
It is a range of 60-150.
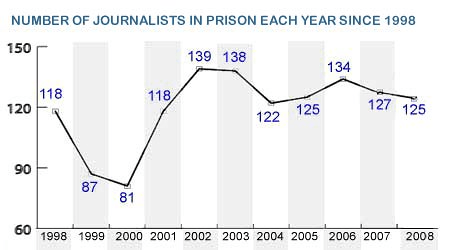
What is the dependent variable?
It ranges from 1998 to 2008.
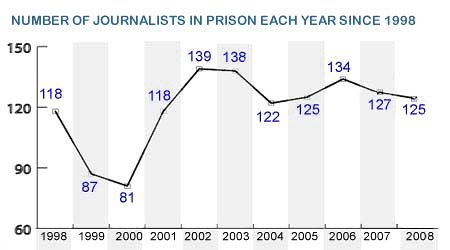
What is the independent variable?
 The months January thru April
The months January thru April
What is the independent variable?
 The range 0-20
The range 0-20
What is the dependent variable?