The unit measure for Speed?
M/s or Miles /h or km/h
what is the density of a cube with mass of 300 g and volume of 3000 cm3?
0.1 g/ cm3
Trial mix is a
a) homogenous
b) heterogenous
c) colloid
d) not known
heterogenous mixture
when an element has different isotopes, it means
a) there are different versions of this element with different # of electrons.
b) there are different versions of this element with different # of protons.
c) there are different versions of this element with different # of neutrons.
there are different versions of this element with different # of neutrons.
Is it an atom or molecule?
NaCl
Molecule
How many liters are in 5612 Milliliters?
we are going small to big, milliliters to liters.
we need to divide.
answer is 5.612 liters.
What is the density of a liquid with volume of 12ml and 120 grams of mass?
10 g/liter
What is a solute?
a substance that get s dissolved.
An isotope of iodine has atomic mass or 123. How many neutrons does it have ?
70
because 123- 53 = 70
How many oxygen atoms are here in this molecule?
2H2SO4
8
how many meters are 5600 millimeters?
5.6 meters
What is the density of a cube that has one of the dimension at 2.5 cm and mass of 1560 grams?
100 gr/cm3
The main difference between a solution and colloid and a suspension is
difference in particle sizes in solution , suspension and colloid
An Isotope of Pb has atomic mass of 209. How many protons it has?
82
Is it a cation or anion or a neutral atom?

Cation
How many kg is 0.00234 milligrams?
0.00000000234. or 2.34 x 10 -9
What is the volume of an object that has mass of 250 g and density of 13.7 gr/cm3?
18.2 cm3
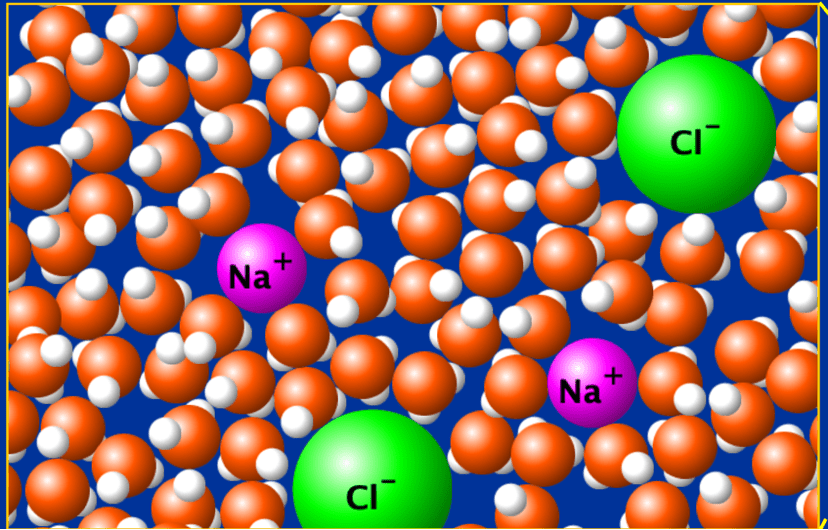
is it a homogenous or heterogenous mixture?
homogenous mixture
Pb-209, how many neutrons it has?
209-82 = 127
This is a density graph of substance A. If substance B has a higher density than substance A, where would you draw a line representing density of substance B? on top or below the line shown ?

c
How many meters are in 0.0234 decimeters ?
0.00234
What is the mass of an object that displaces 420 milliliter of water and has density of 10 g/ml?
4200 g
is air
a) compound
b) mixture
c) element
mixture
If an isotope of Carbon has 6 protons and 8 neutrons, what is atomic mass of this type of Carbon?
14
Is one kg of feather is more dense or one kg of bricks?
One Kg of Bricks