WHICH NATURALLY OCCURING RADIOACTIVE POLLUTANT IS A MAJOR CAUSE OF LUNG CANCER?
Radon
NAME TWO FACTORS THAT INCREASE PHOTOCHEMICAL SMOG FORMATION
Strong sunlight
High concentrations of nitrogen oxides (NOₓ) and volatile organic compounds (VOCs)
CO
Carbon Monoxide.
Motor vehicle exhaust
Incomplete combustion of fossil fuels (cars, furnaces, generators)
Residential heating and cooking (gas, oil, wood)
 Identify this disease.
Identify this disease.
Pulmonary edema
WHY DID ATMOSPHERIC LEAD LEVELS DROP DRASTICALLY IN THE 70’s?
The phase-out of leaded gasoline
WHAT IS ONE CAUSE OF NOISE POLLUTION AND ONE HARMFUL EFFECT?
Cause: Traffic or aircraft noise
Harmful effect:
Hearing loss, sleep disturbance, disruption of migration patterns.
WHAT IS PM2.5 AND WHY IS IT SO DANGEROUS?
PM₂.₅ is fine particulate matter with a diameter of 2.5 micrometers or smaller.
It is dangerous because these particles can be inhaled deep into the lungs and enter the bloodstream, causing respiratory and cardiovascular disease.
EXPLAIN TWO REASONS FOR THE URBAN HEAT ISLAND EFFECT
Dark, impervious surfaces (asphalt, concrete) absorb and retain heat.
Lack of vegetation reduces cooling from shade and evapotranspiration
O3
Photochemical reactions between nitrogen oxides (NOₓ) and volatile organic compounds (VOCs) in sunlight
IDENTIFY

WHICH INVENTION, NOW REQUIRED ON ALL U.S. VEHICLES, IS CREDITED FOR REDUCING NO2 AND HENCE SMOG?
The catalytic converter.
WHAT ARE TWO WAYS TO REDUCE THE RISK OF CO POISONING IN YOUR HOME?
Install carbon monoxide (CO) detectors
Properly ventilate and maintain fuel-burning appliances
WHERE WOULD YOU FIND “BAD” OZONE AND WHERE DOES IT COME FROM?
“Bad” ozone is found in the troposphere (near ground level).
It comes from chemical reactions between nitrogen oxides (NOₓ) and volatile organic compounds (VOCs) in the presence of sunlight.
WHAT IS THE ROLE OF VOCs IN PHOTOCHEMICAL SMOG FORMATION?
VOCs react with nitric oxide (NO), preventing NO from breaking down ozone (O₃), which allows ozone to accumulate in photochemical smog.
VOCs
Vehicle exhaust
Industrial solvents and chemical manufacturing
Evaporation of fuels (gasoline, oil)
Paints, aerosols, and cleaning products
Identify this device:
Electrostatic precipitator
NAME A SOURCE OF PM POLLUTION AND A WAY TO REDUCE IT
Coal-fired power plants → Electrostatic precipitators or scrubbers
Vehicle exhaust → Catalytic converters and reduced vehicle use
Construction and road dust → Water spraying and vegetation ground cover
IDENTIFY THE TOP THREE MOST ABUNDANT GASES IN THE ATMOSPHERE WITH THEIR APPROXIMATE PERCENTAGES
Nitrogen (N₂) — ~78%
Oxygen (O₂) — ~21%
Argon (Ar) — ~0.9%
WHICH TWO PRIMARY POLLUTANTS EVENTUALLY BECOME ACID PRECIPITATION AND WHAT IS THE PRIMARY SOURCE OF EACH ONE?
Sulfur dioxide (SO₂) — primarily from burning coal in power plants
Nitrogen oxides (NOₓ) — primarily from motor vehicle exhaust (also fossil-fuel combustion in power plants)
IDENTIFY TWO CITIES THAT ARE AT HIGH RISK OF PHOTOCHEMICAL SMOG
Los Angeles
Mexico City
Also: Beijing, Delhi, Houston, Phoenix
Pb
Lead:
Coal combustion
Metal smelting and processing
Leaded gasoline (historically)
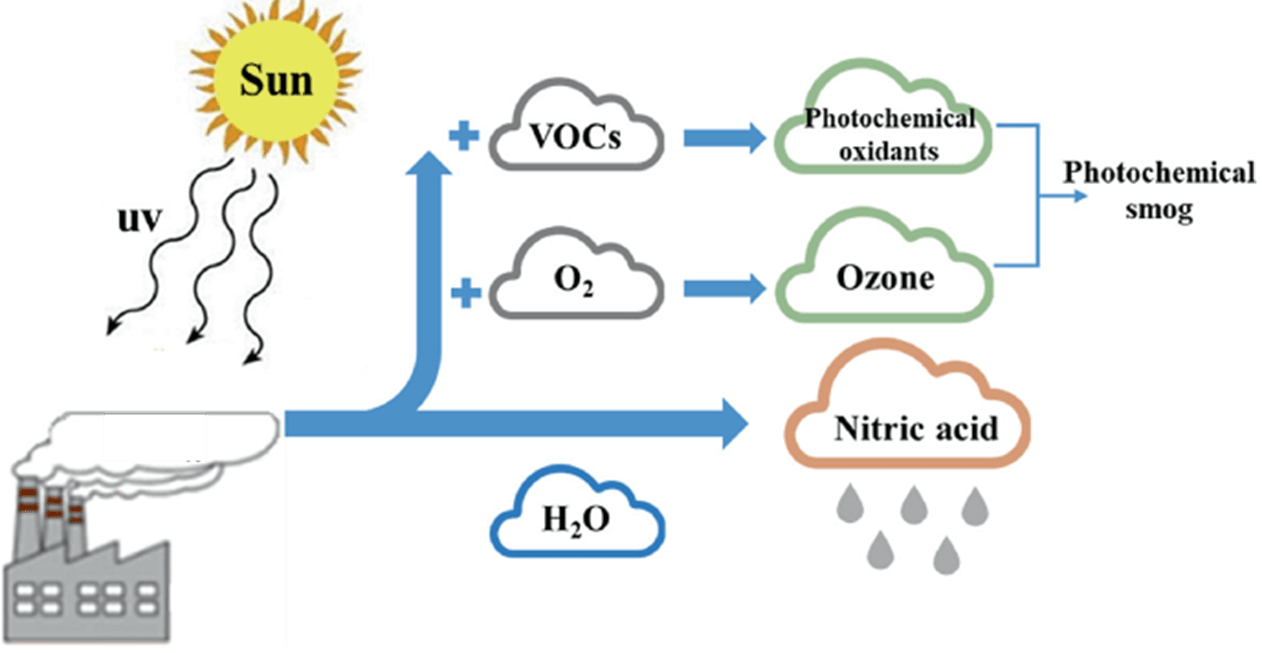
What pollutant is coming from the power plant?
Nitrogen oxides (NOx)
WHICH PIECE OF TECHNOLOGY CAN MOST EFFECTIVELY REDUCE SO2 EMISSIONS FROM COAL-FIRED POWERPLANTS?
Scrubbers (flue-gas desulfurization systems)
EXPLAIN THE PROBLEM SHOWN IN THIS DIAGRAM
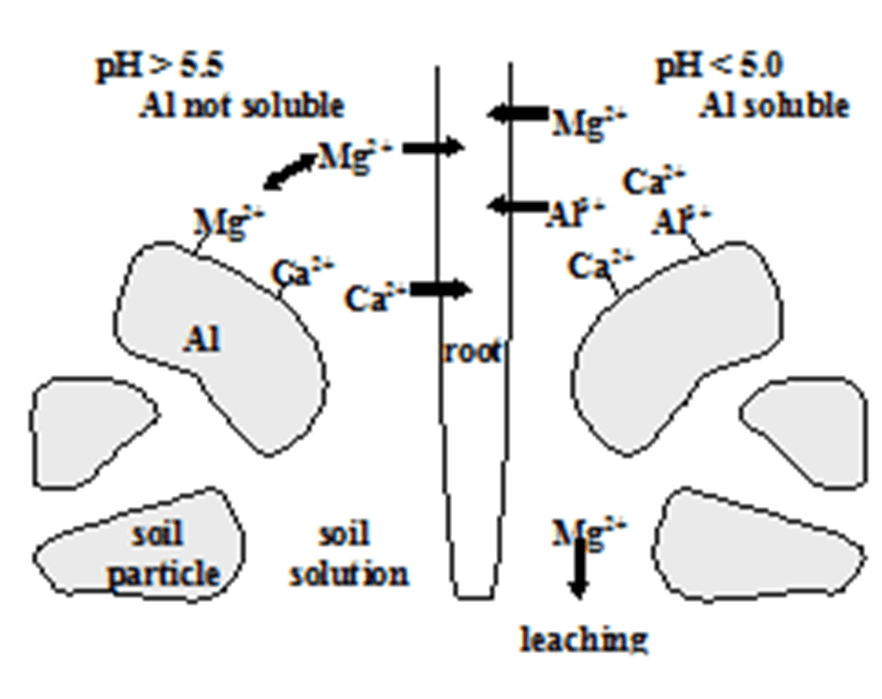
Soil acidification (from acid deposition)
As soil pH drops below ~5, aluminum (Al³⁺) becomes soluble and is released from soil particles. This toxic aluminum damages plant roots and displaces essential nutrients like calcium (Ca²⁺) and magnesium (Mg²⁺), which are then leached away.
Result: nutrient loss + root damage → reduced plant growth and forest decline.
NAME THREE DIFFERENT POLLUTANTS THAT ARE PRODUCED BY NATURE AND THEIR RESPECTIVE SOURCE
Radon — released from the natural decay of uranium in rocks and soil
Volcanic ash / sulfur dioxide (SO₂) — emitted during volcanic eruptions
Pollen — produced by plants
IDENTIFY THE REGIONS OF WARM, COOL, AND COOLER AIR IN EACH DIAGRAM (Don’t go by the colors)
Normal Diagram:
1. Cooler Air
2. Cool Air
3. Warm Air
Thermal Inversion:
1. Cool Air
2. Warmer Inversion Layer
3. Cool Air
H2SO4
Sulfuric Acid
Coal combustion
Oil combustion
Atmospheric reactions involving sulfur dioxide (SO₂)
IDENTIFY THIS PHENOMENON
WHAT DEVICE CAN DRASTICALLY REDUCE VOCs FROM GASOLINE DURING REFULING?
Vapor recovery nozzles (Stage II vapor recovery systems)
EXPLAIN THIS GRAPH
This graph shows photochemical smog formation over the course of a day.
Morning: NO is high from vehicle emissions and destroys ozone, keeping O₃ low.
Midday (sunlight increases): NO is converted to NO₂ by reactions with oxygen and VOCs; NO₂ peaks.
Afternoon: Sunlight breaks NO₂ apart, allowing ozone (O₃) to accumulate and peak.
Evening: Sunlight decreases, ozone production drops, and NOₓ levels decline.