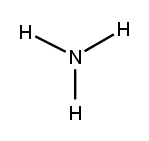
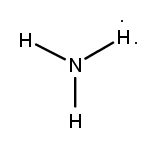
What type of bond forms when electrons are shared?
covalent bonds
What is the name of the following monatomic ion?
Fe3+
iron (III) ion
Water
What is the number for N for the lewis structure of N2?
What is 16?
Which element on the periodic table has the highest electronegativity?
What type of bonds are formed when electrons are transferred?
ionic bonds
What is the name of this ionic bond?
CuO
copper (II) oxide
What is the name? N2O5
dinitrogen pentoxide
How many bonds are formed in the following compound? SiO2
4 bonds
2 - double bonds
Which type of bond has a electronegativity of less then 0.4?
nonpolar covalent bond
A ________ ion gains elctrons?
nonmetal
What is the name of the following:
BaCl
barium chloride
Name H2S
hydrosulfuric acid
How many lone pairs are present in the lewis structure of N2?
2 lone pairs
What type of bond is N-O?
polar covalent
Covalent bonds are formed between _________ and _______ and also _________ and __________.
nonmetal and nonmetal
nonmetal and metalloid
What is the formula for lead (IV) sulfate?
Pb(SO4)2
Name the covalent bond? NH3
ammonia
not nitrogen trioxide
must know common name
Which of the following is the correct Lewis structure of NH3?
Which element on the periodic table has the lowest eletronegativity?
francium or cesium
What determines the identify of an element?
proton
SnN
Tin (III) nitride
Name both the following: H2SO4 H2SO3
Sulfuric acid
Sulfurous acid
Draw the lewis structure of CO2?
O=C=O
two lone pairs on the oxygens.
Which type of bond has the greatest difference in electronegativity?
ionic bond