What are the subatomic particles found in each atom?
Protons, neutrons, and electrons
What does the atomic number of an element tell us?
# of Protons and electrons
What are valence electrons?
Electrons in the outermost shell
What does the coefficient tell us?
The number of molecules
How many hydrogen and oxygen atoms are in 2H2O?
H: 4
O: 2
What charge do protons, neutrons, and electrons have?
+, 0, -
What does the atomic mass of an element tell us?
Total # of protons + neutrons
True or False? Lewis Dot structures show electrons from every electron shell
False
What does the subscript tell us?
The number of atoms within each molecule.
How many molecules are in C6H12O6?
1
What is magic number for valence electrons?
8
How do you find out how many valence electrons are in an element?
Look at the element's group number.
Explain the Octet Rule
Ex) Atoms are most stable when they have a full outer shell of valence electrons
Atoms will gain or lose electrons to fulfill Octet Rule
Atoms will attempt to fill outer shell by gaining or losing the fewest electrons possible
Is 2Fe2O3 an element, molecule, and/or compound?
Molecule and compound
How many atoms in total are in 4K3PO4?
32
What are four of the most common elements in life?
CHON
What are the names of Group 1, 17, and 18?
Group 1: Alkali
Group 17:Halogens
Group 18: Noble gases
Draw a Lewis Dot Structure for Phosporous
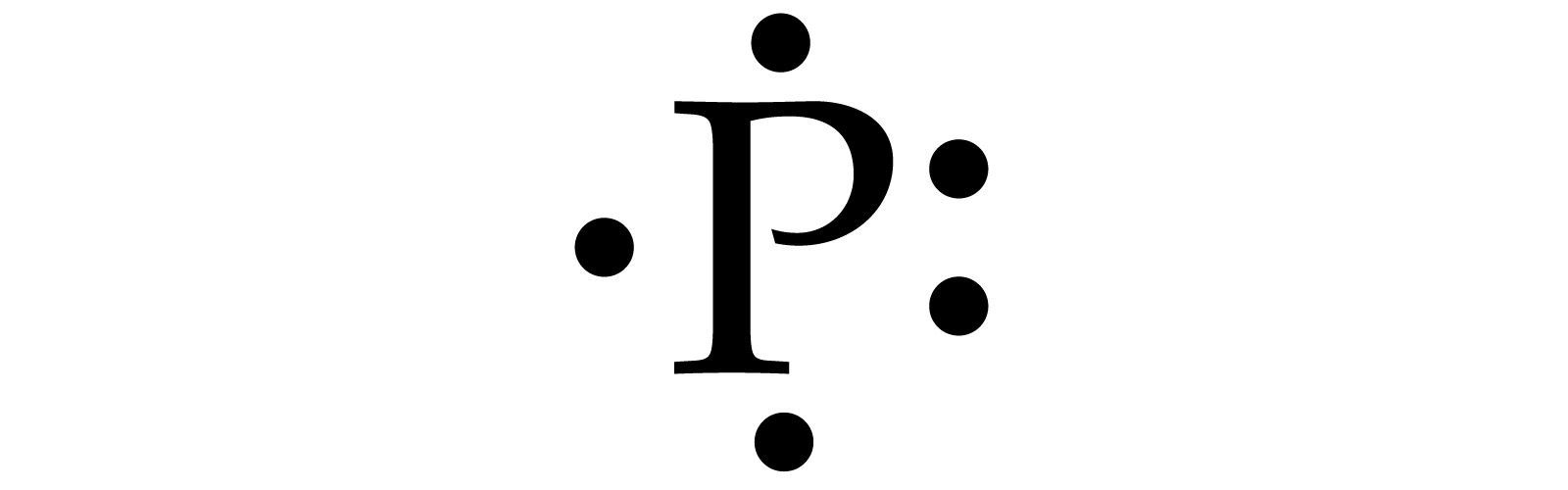
What are the elements, subscripts, and total number of atoms in NaHCO3?
Elements: Sodium, Hydrogen, Carbon, Oxygen
Subscripts: 3 (hidden subscript is 1)
Number of atoms: 6
Draw 3H2O (color code or write out the element symbol in a circle).
Should be 3 separate molecules of H2O
How many protons, neutron, and electrons are in Ytterbium?
70, 103, 70
At what atomic number and above are elements too unstable to occur in nature?
92 (uranium)
Draw a Lewis Dot Structure for Selenium. Also explain if it would gain or lose electrons and how many?
It would gain 2 electrons because it is the fewest amount of electrons to be gained or lost
How many elements are in Na2CO3? How many atoms of each element are in Na2CO3? How many molecules are there in Na2CO3?
3 ; Na, C, O
Na = 2 C = 1 O = 3
1 molecule
Draw CO2 + H2O (color code or write out the element symbol in a circle).