True/False: When liquid water changes to a solid, the particles slow down and move closer together because energy is removed from the water.
True- when heat energy is removed, particles slow down.
Is this a heterogenous or homogenous mixture? WHY?

Heterogenous because it is not evenly mixed, it has visible, separate parts, and is easy to separate
True or false?
Mixtures are physical changes and can be separated.
True!
This type of change only changes properties like shape, size, or state of matter. It is usually reversible, new substances are not formed. What type of change is this?
A physical change.
True/False: When water changes from a liquid to a gas, the particles move faster and spread farther apart because the water gains energy.
True! When heat energy is added, particles speed up and spread farther apart.

Is this a heterogenous or homogenous mixture? WHY?

Homogeneous mixtures (like salt water or juice) are perfectly blended, look the same throughout, and cannot be easily separated.
A student accidentally spills a mixture of sand and iron filings on a table. Which method would best separate the iron filings from the sand?
A. Adding water to dissolve the iron filings
B. Using a magnet to pull out the iron filings from the sand
C. Heating the mixture until the iron filings evaporate
D. Blowing on the mixture to remove the iron filings

B. Using a magnet to pull out the iron filings from the sand

Adding sugar to water- is this a physical or chemical change?

A student leaves a tray of water outside on a hot day. After a few hours, most of the water is gone. Which best explains what happened to the water particles?
A. They stopped moving and became heavier.
B. They moved faster and spread apart into the air.
C. They turned into solid particles in the air.
D. They stayed the same but were squeezed together.
B. They moved faster and spread apart into the air.
The water evaporated! It changed from a liquid to a gas.
Is air a heterogenous mixture or homogenous mixture?

Air is a homogenous mixture of different types of gases. Homogeneous mixtures (like salt water or juice) are perfectly blended, look the same throughout, and cannot be easily separated.
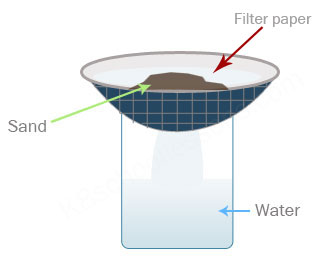
What is the best way to separate this mixture of sand and water?
Use a filter or sieve
One student below created a chemical change, who was it?

Student Y baked cookies- this is a chemical change.
A student notices that a puddle on a warm, windy day dries up faster than a puddle on a cool, calm day. Which explanation uses particle motion and temperature to support a claim about this difference?
A. Wind and warmth add energy, causing particles to move faster and escape into the air as gas.
B. Wind removes particles so they no longer exist.
C. The cool day changes water into a new kind of substance.
D. The puddle on the cool day has no particles.
A. Wind and warmth add energy, causing particles to move faster and escape into the air as gas.
Susie made some hot chocolate like the one pictured below: 
After making her perfect hot chocolate, she decided to add some marshmallows on top. What type of mixture is the hot chocolate before and after the marshmallows?
The hot chocolate was a homogenous mixture, but when Susie added marshmallows, it became a heterogenous mixture.

A student wants to separate a mixture of rice and dried beans. Which method is most practical and effective?
A. Sorting them by hand based on size and shape
B. Dissolving them in water and then evaporating the water
C. Using a magnet to pull the rice away
D. Burning the mixture and keeping what is left
A. Sorting them by hand based on size and shape
Is this a chemical or physical change?

Cooking or burning is a chemical change- it creates a new substance that is not reversible. Clues of a chemical change are bubbles, a color change, a new smell, or light.
Part 1: Which state of matter has a definite shape and a definite volume?
Part 2: Which state of matter has a definite volume but not a definite shape?
Part 3: Which state of matter does not have a definite shape or volume?
Part 1: Solid
Part 2: Liquid
Part 3: Gas
Oil and water are both liquids. When they are mixed together in a cup, what type of mixture is this? Why?

Oil and water do not mix well, they are a heterogenous mixture.
A student wants to show that a mixture can be separated using differences in particle size. Which investigation best demonstrates this concept?
A. Separating a saltwater solution by evaporation
B. Separating a mixture of sand and pebbles using a screen with holes smaller than the pebbles
C. Separating iron filings from sand with a magnet
D. Separating oil and water by pouring off the top

B. Separating a mixture of sand and pebbles using a screen with holes smaller than the pebbles
Rosie is preparing a salad using the following steps:
1. Cutting up lettuce
2. Cutting tomatoes
3. Adding oil and vinegar
4. Boiling an egg
Classify each step as a physical or chemical change.
1. Cutting up lettuce - PHYSICAL
2. Cutting tomatoes - PHYSICAL
3. Adding oil and vinegar - PHYSICAL
4. Boiling an egg - CHEMICAL
