Is the formation of chemical Bonds Endothermic or Exothermic?
Exothermic
Draw the structure for pentane
What phase (or phases) are present during segment A?
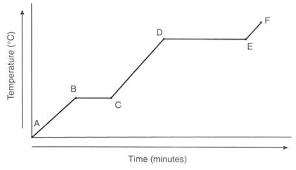
Solid
How much heat is required to raise the temperature of 100 g of water from 20°C to 50°C?
12,540 J
Why is more energy required for vaporization than for fusion?
When particles go from a liquid → gas, they must separate completely and move far apart, which requires breaking most or all intermolecular forces.
What is the transition phase from solid to gas?
Sublimation
Is water evaporating endothermic or exothermic?
Endothermic: energy is being absorbed by the water
Draw this organic compound: C4H8

What phase change, if any, is occurring during segment DE?

Liquid to gas
How much heat is required to melt 50 g of ice at 0°C?
ΔHfus= 334 J/g
16,700 J
The lowest temperature that is theoretically possible, at which motion of particles that constitutes heat would be minimal
Absolute Zero, 0 Kelvin, or -2730C
What is the transition phase from gas to solid?
Deposition
I thi Reaction Endothermic or Exothermic?

Exothermic
Calculate the bond enthalpy for the reaction.
Is the reaction endothermic or exothermic?
678 - 862 = ~ -184 kJ/mol
Exothermic
What phase (or phases) are present during segment BC?

Solid and Liquid
How much heat is required to convert 60.0 g of water at 25.0°C into steam at 110.0°C?
ΔHfus= 334 J/g
ΔHvap= 2260 J/g
155,616 J
The specific temperature and pressure at which a substance exists in all three phases at the same time: solid, liquid, and gas.
Triple Point
At 30 atmospheres pressure, the boiling point of this substance is:

50 ºC
Calculate the energy, in Joules, that would be released if you ate 104 g of this food

502,000 J
Calculate the bond enthalpy of this reaction

6494 - 8528 = ~-2034 kJ/mol
At what segments does the latent heat of fusion occur?

CB
How much total heat is required to convert 75.0 g of ice at −15.0°C into steam at 120.0°C?
ΔHfus= 334 J/g
ΔHvap= 2260 J/g
231,000 J
The specific temperature and pressure at which the distinction between a liquid and a gas disappear
Critical Point
A phase change from Phase B to Phase C is known as:

Freezing
Draw the potential energy diagram (graph) for an exothermic reaction, label Enthalpy (ΔH) and Activation energy (Ea)

Calculate the enthalpy of combustion of ethane using bond enthalpies
2C₂H₆ + 7O₂ → 4CO₂ + 6H₂O
9117 - 11948 = ~ -2800 kJ/mol
At what segments does the latent heat of vaporization occur?

DE
How much total heat is required to convert 125 g of ice at −25.0°C into steam at 130.0°C?
ΔHfus= 334 J/g
ΔHvap= 2260 J/g
390,569 J
How old is Cricket?
2
The triple point of this substance occurs at:
- 15 ºC and 6 atmospheres