The tiny pieces that all matter is made of.
Atoms
What was the brown substance in the water in Westfield?
Rust
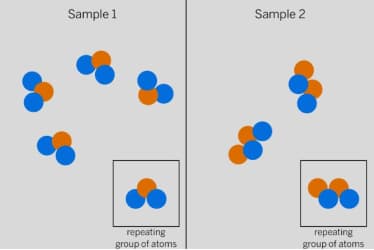
These substances will have __________ properties.
different
A starting substance of a Chemical Reaction
What is a REACTANT
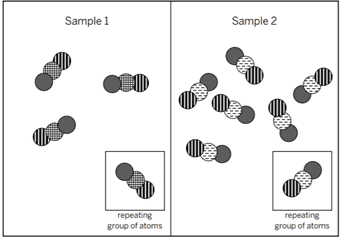
The diagram shows the repeating groups of atoms that make up two samples. The properties will be ________ because ________.
different; the molecules are made of different types of atoms
Something that is all the same atoms or groups of atoms.
What is a SUBSTANCE
Could the rust have been created without fertilizer in Westfield's pipes?
No.
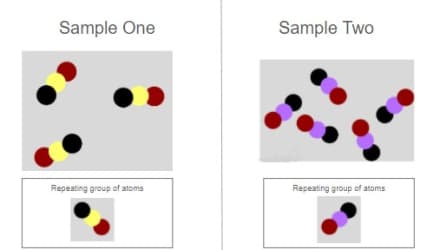
These substances have the exact same properties. True or False?
False
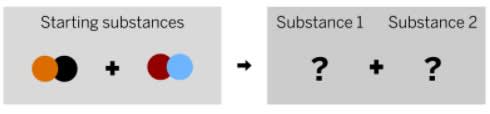
Can these reactants form this product?

no
In a balanced chemical equation, why must the number of atoms for each element be the same on both sides of the equation?
What is To satisfy the Law of Conservation of Mass, indicating that atoms are neither created nor destroyed in a chemical reaction.
A process in which atoms rearrange to form new substances
Chemical Reaction
What two substances created rust?
Fertilizer and Iron
A group of atoms joined together in a particular way.
What are MOLECULES
In the modern periodic table, elements are arranged according to their
What is ATOMIC NUMBER and ATOMIC MASS


Which of the diagrams to the left shows the repeating groups of atoms that make up the ending substances?
C
An ending substance that is made during a chemical reaction.
Product
Why didn't the neighboring town have rust?
There was no fertilizer in the water.
What can cause an chemical reaction?
When molecules are able to combine using - liquid, heat, cold, or mixing
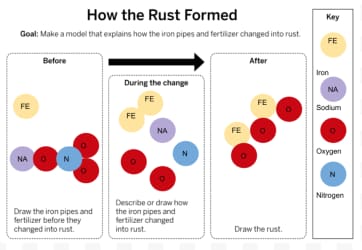
Can substances change into different substances?
yes, because atoms can be rearranged to form different substances
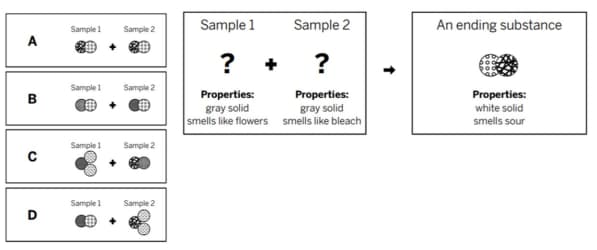
Which of the diagrams shows the repeating groups of atoms that make up the reactants?
A
Something that can be observed about a substance, such as color, smell or boiling point.
What is a PHYSICAL PROPERTY
How was rust created? Which atoms were involved?
A chemical reaction caused iron and oxygen to combine from the fertilizer running through the pipes
In this chemical equation, which are the products? 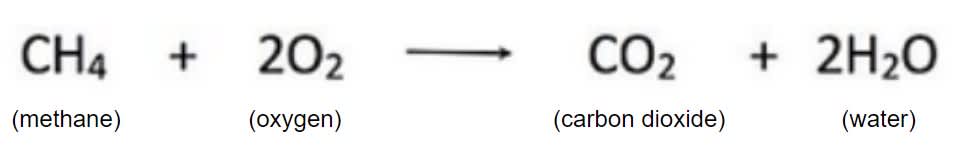
Water and carbon dioxide
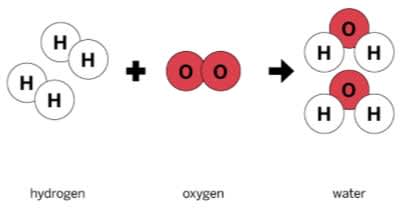
In the reaction shown, hydrogen and oxygen are the _______.
Reactants
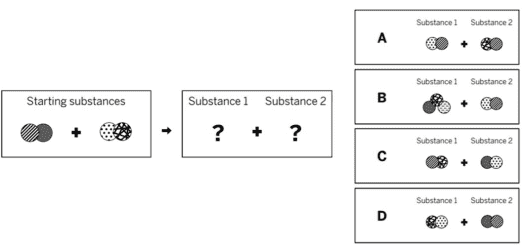
Which of the diagrams to the right shows the repeating groups of atoms that make up the products?
c
