The tiny pieces that all matter is made of.
Atoms
What was the brown substance in the water in Westfield?
Rust
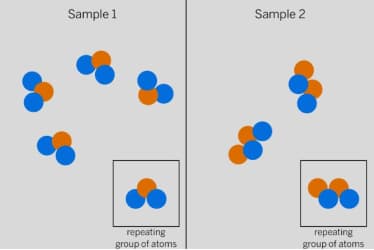
These substances will have __________ properties.
different
The temperature at which a substance changes from the solid phase to the liquid phase is
Melting point
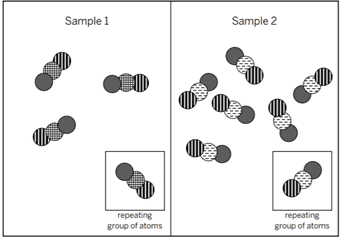
The diagram shows the repeating groups of atoms that make up two samples. The properties will be ________ because ________.
different; the molecules are made of different types of atoms
An object, diagram, or computer program that helps us understand something by making it simpler or easier to see.
Model
What Chemical compound did you observe that has the generic name Rust
Iron Oxide
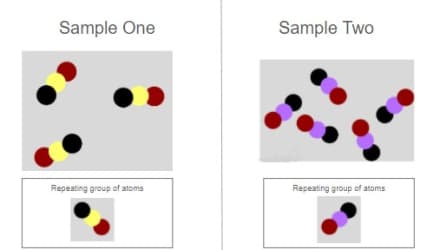
These substances have the exact same properties. True or False?
False
What is the name for the starting substance of a chemical reaction
Reactants
During a chemical reaction, atoms cannot be ______ or ______.
Created or destroyed
A process in which atoms rearrange to form new substances
Chemical Reaction
What two substances created rust?
Name three properties scientist observe to tell chemicals apart.
color, smell, texture, phase, and melting/boiling point,
What was the chemical name for the scent added into fruit juice?
Ethyl butyrate
Name of the Scientist that Discovered the Law of Conservation of Matter
Antoine Lavoisier
An ending substance that is made during a chemical reaction.
Product
Fertilizer
Adding liquid, heat, cold or mixing a substance can cause what?
A Chemical Reaction
What was the chemical name for the substance that smells like sweaty socks?
Isovaleric Acid
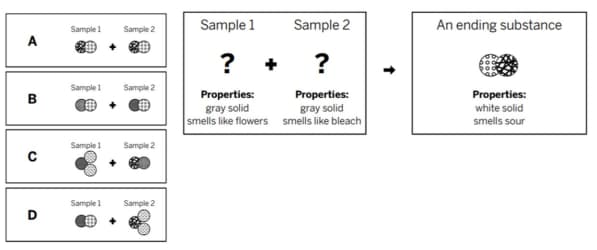
Mr. Cosgrove mixed two samples together: a gray solid that smells like flowers and a gray solid that smells like bleach. He analyzed the results and found two ending substances. One of the ending substances (shown in diagram) was a white solid, made up of the repeating group of atoms shown above. Which of the diagrams shows the repeating groups of atoms that make up the reactants?
D
Something that can be observed about a substance, such as color, smell or boiling point.
Property
What two substances were revealed to be in Westfield's Water
Rust and Sodium Nitrite
In this chemical equation, which is one of the products? 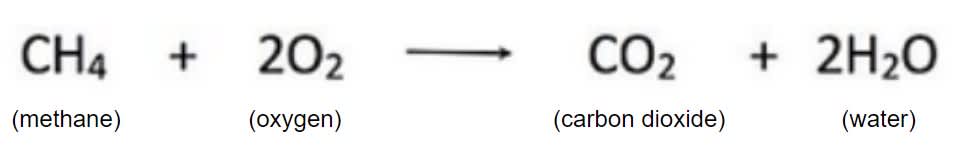
Water
Carbon Dioxide
_______ is a measure of how much matter makes up an object
Mass
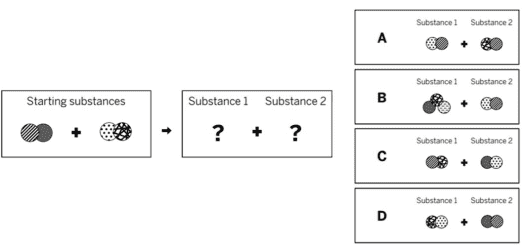
Ms. Schoolcraft is a chemist who is making a chemical to add to swimming pools in order to make the water safer. She mixed two solid substances together in a sealed container. The diagram above shows the repeating groups of atoms that make up the two starting substances. After mixing, Ms. Schoolcraft found two liquid substances in the sealed container. (Nothing had escaped.) Which of the diagrams to the right shows the repeating groups of atoms that make up the products?
c
