an arrangement of the elements by increasing atomic number and by changes in physical and chemical properties.
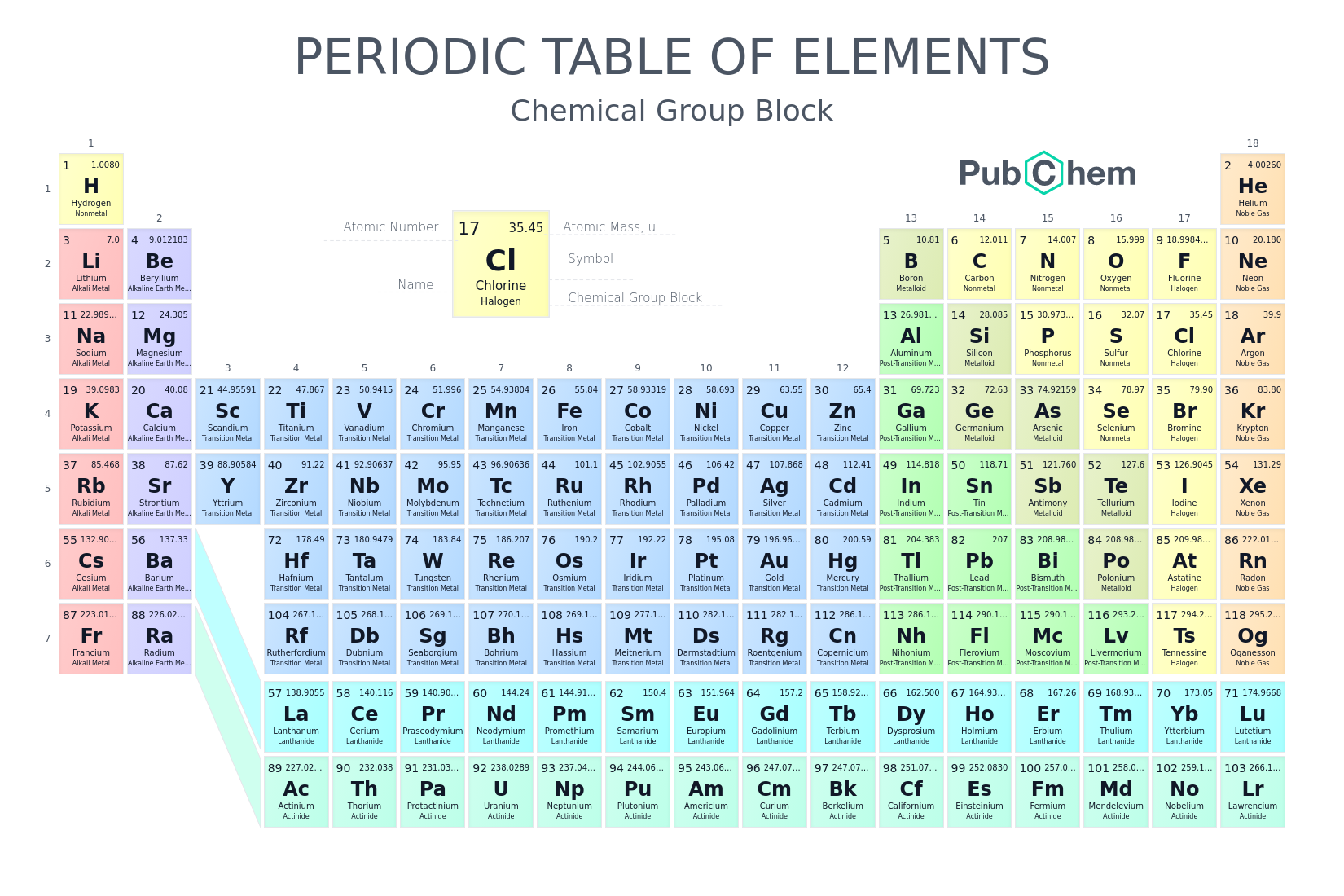 periodic table
periodic table
given an atomic number, 16 how many protons or electrons does it have?
16
given an atomic number, how many neutrons does it have? 20
20 neutrons (Calcium)
Na on the periodic table
What is Sodium
how does static electricity work? (what part of an atom moves?)
electrons (the negatively charged particles in an atom) move from one material to another.
what atom Is this
Zinc
which element is atomic number 12?
magnesium
Given a picture of an atom, can you tell which element it is? 
Boron
What is the atomic mass of an atom equal to?
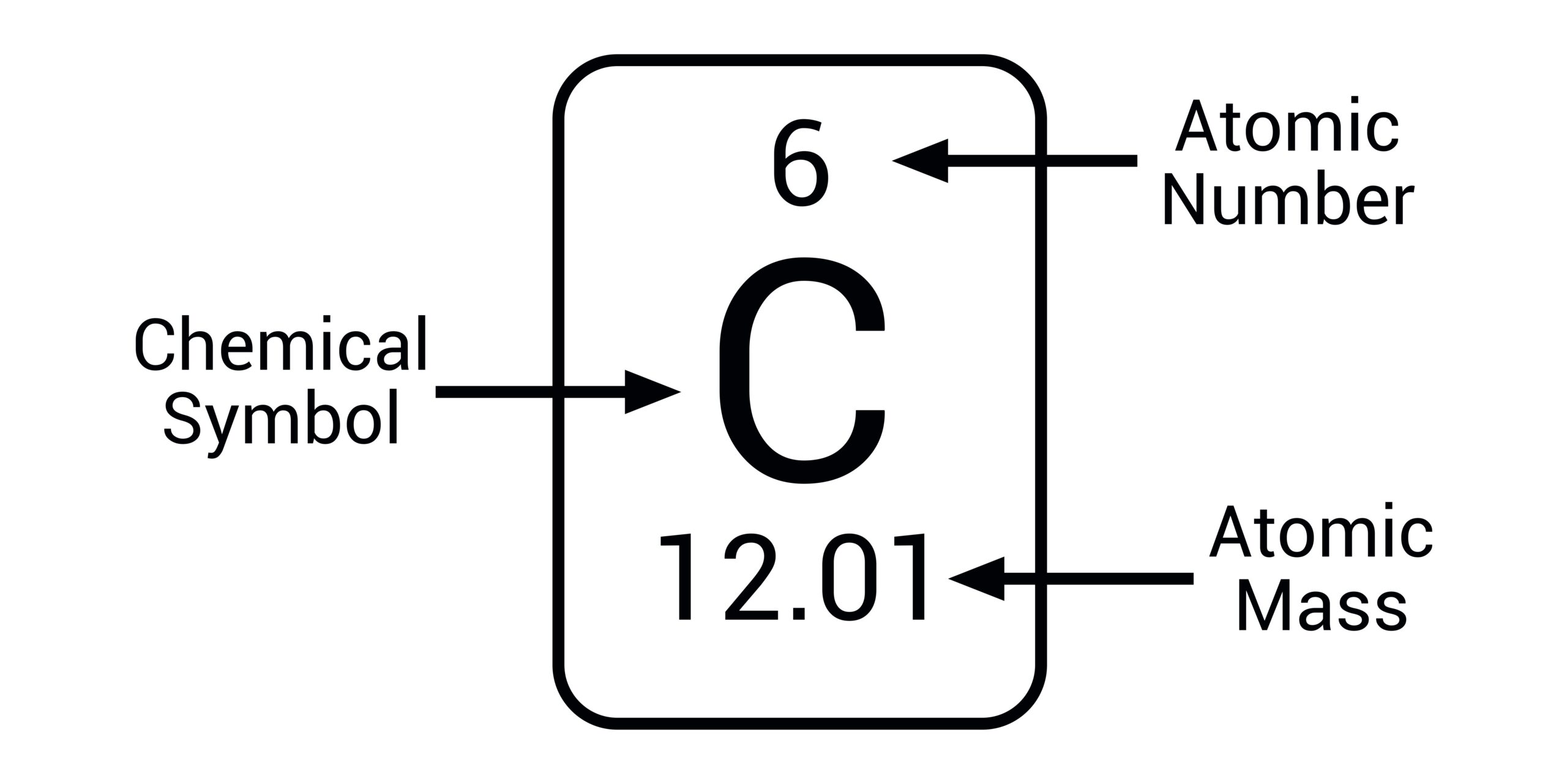 the number of protons plus the number of neutrons that it contains.
the number of protons plus the number of neutrons that it contains.
What do the numbers on the top of the element mean?
Atomic number

what atomic number would this atom be?
6 (carbon)
True or False. Do the colors on the periodic table of elements mean anything?
True. Periodic tables can use color to identify other element characteristics.
What is a molecule, and how would you recognize one?
A molecule is two or more atoms connected by chemical bonds, which form the smallest unit of a substance that retains the composition and properties of that substance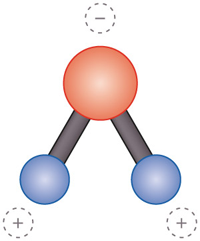
What is the name of element number 1?
Hydrogen.
What is Irons atomic symbol?
FE
Who had the thought of elements
Democritus
the sum of the number of protons and the number of neutrons in an atom
What is the mass number
the area around the nucleus of an atom where its electrons are most likely found
What is an electron cloud