__________ are the basic building block of all matter.
What are atoms?
The number of protons in an atom of an element is its ___________ number.
What is atomic?
The GROUP this element is found in
13
A positively charged subatomic particle.
What is a proton?
All of the rows have this in common
What are number of energy levels?
This type of element is KIND OF brittle but KIND OF shiny, KIND OF
Metalloids?

All of the "blue" elements are considered to have what similar characteristic (or property)
What is a metal?
How many valence electrons in group 18?

2 Helium, 8 all others
When looking at the periodic table, the vertical columns all have similar what?
What is a property?
The sum of protons and neutrons in the nucleus.
What is : atomic mass
Electrons are found here.
What is the electron cloud?
Rows of elements are called this.
What are Periods?
The atomic weight of this element.
What is 55.845
A negatively charged subatomic particle
What is an electron?
What's the matter?
EVERYTHING THAT TAKES UP SPACE
In an atom, which has the least mass?
A: nucleus
B: proton
C: neutron
D: electron
What is D: electron
Name 3 characteristics of a an element that is a METAL
What is ductile, shiny, good conductors?
A carbon atom with a 7 neutrons and 6 protons would have what mass number?
Bonus: write it in isotopic notation
13
Elements found in the same group (column) have the same..
The answer, my good sir, is the same PROPERTIES and the same VALENCE ELECTRON NUMBER?
A fluorine atom has 9 protons, 10 neutrons, and 9 electrons. What is the atomic number?
9
Protons and neutrons are found here.
What is the nucleus.
The number that tells us the total number of protons plus neutrons at the bottom of each element square on the periodic table.
What is the atomic mass number?
The number of neutrons in this element.
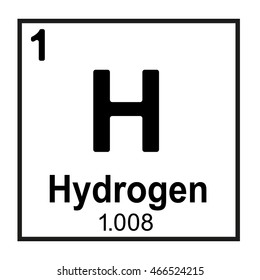
What is zero?
An atom that has a different number of neutrons
What is an isotope
This element has 32 protons and an atomic mass of 95. How many neutrons will it have?
What is 63 neutrons?
What is an atom mostly made of?
empty space
What are electrons called in the outer shell/ energy level
valence electrons
Is this element a metal, metalloid, or nonmetal

Metal
The overall charge of a whole atom is this.
What is zero.
What determines the identity of an element.
What is the number of protons or atomic number?
