What is the element with the atomic number of 30?
Zinc
The number of electrons is the same as the number of:
Protons
Neutron is _______ charged
Uncharged/ neutral
Nitrogen
7
What does a nucleus contain?
Protons and neutrons
 How many atoms of carbon are in this molecule?
How many atoms of carbon are in this molecule?
1
How many protons and electrons does this element have? Atomic number: 4
4 protons, 4 electrons
The positively charged particle
Proton
electrons are _____ charged
Negatively
Iron
30
True or False? The material that gains electrons becomes negatively charged.
True
How much helium does this molecule have?
How many neutrons does an element with an atomic number of 21?
About 24 (23.956)
The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the _____
Atom
Atoms with an unusual amount of neutrons are called _______________
isotopes
Xenon
77
 Are electrons inside or outside the nucleus?
Are electrons inside or outside the nucleus?
outside
How much oxygen is in this molecule?
1
What is the atomic mass of a molecule with the atomic number of 85?
210
In an atom, which part has the least mass?
Electron
The sum of protons and neutrons in the nucleus is the
Atomic mass
Strontium
50
What part of an atom can move around from one atom to another?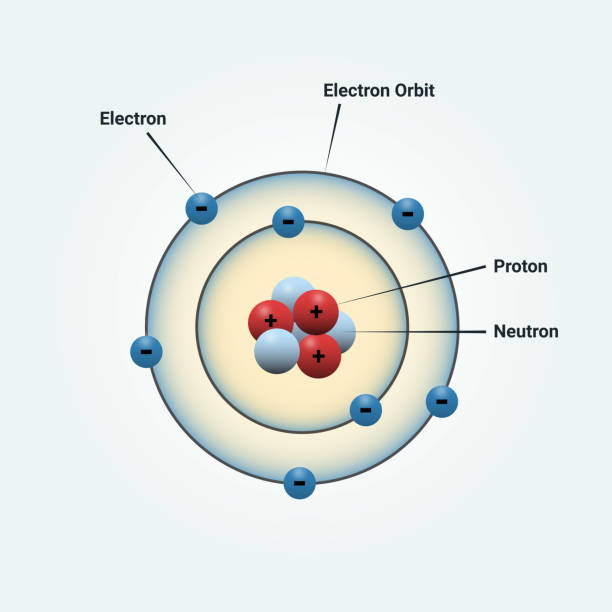
Electrons
What element is this?
Hydrogen
Uranium-235 has this many neutrons.
What is 143?
Which is bigger; the proton or the electron?
The isotope of iodine 130 has _________ more neutrons than atomic iodine.
3 more!
Atomic iodine has 127-53 = 74 neutrons
Iodine 130 has 130-53 = 77 neutrons.
Ununpentium
171
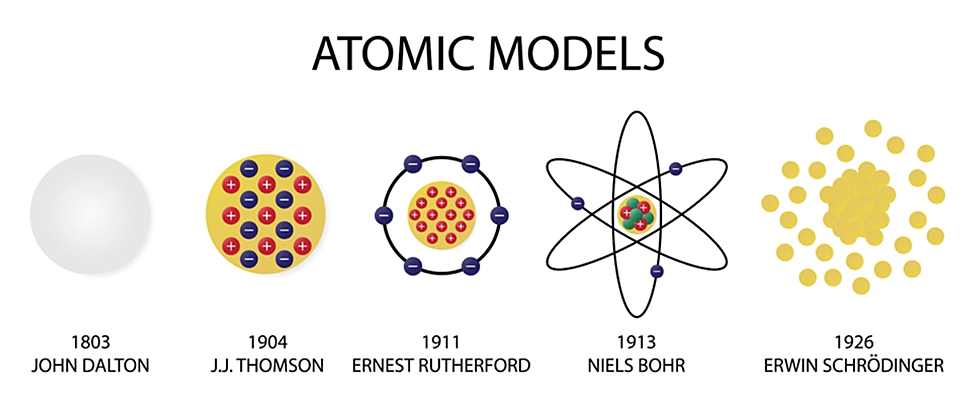 What is a molecule and how do you recognize one?
What is a molecule and how do you recognize one?
Atoms bonded together to make a new substance with unique physical and chemical characteristics.
What element is this?
Carbon