What charges to protons, neutrons, and electrons each have
Protons - positive (+)
Neutrons - neutral (0)
Electrons - negative (-)
What is an element?
An element is a pure substance made of only ONE type of atom
What is a Molecule? Give an Example.
Two or more atoms bonded together.
Examples may vary.
How do you calculate the number of neutrons in an atom?
a. Same as the number of Protons
b. Atomic Number - Electrons
c. Atomic Number - Atomic Mass
d. Atomic Mass - Atomic Number
Atomic Mass - Atomic Number
What is Ms. Reed's favorite animal?
What are dogs.
Inside an atom there is mostly...... what?
What is Empty Space!
Potassium's Atomic Number is 19. Its Atomic Mass is 39.0983.
How many neutrons are in a Potassium Atom?
20 neutrons
This is a molecule of two hydrogen atoms (H2). What part of an atom determines how it bonds to others?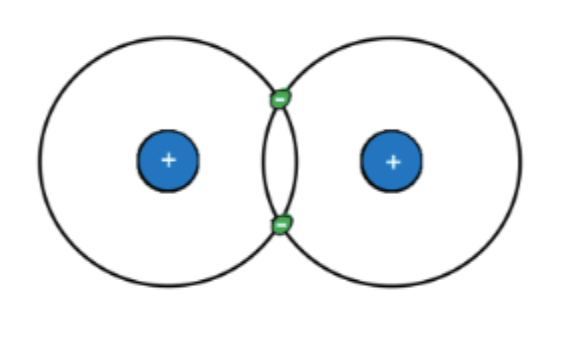
Electrons.
An atom's electrons (number, arrangment) determines how it will or won't bond with other atoms.
How many protons, electrons, and neutrons does Sodium have in its atoms?

11 protons
11 electrons
12 neutrons
What state was Ms. Reed Born in?
What is Maine.
How many electrons can go in the first electron cloud/shell?
what is 2.
The weights of atoms, protons, neutrons, and electrons is measured in AMUs.
What does AMU stand for?
Atomic Mass Unit
This is a Carbon atom. How many more electrons does it need to have a full outer shell?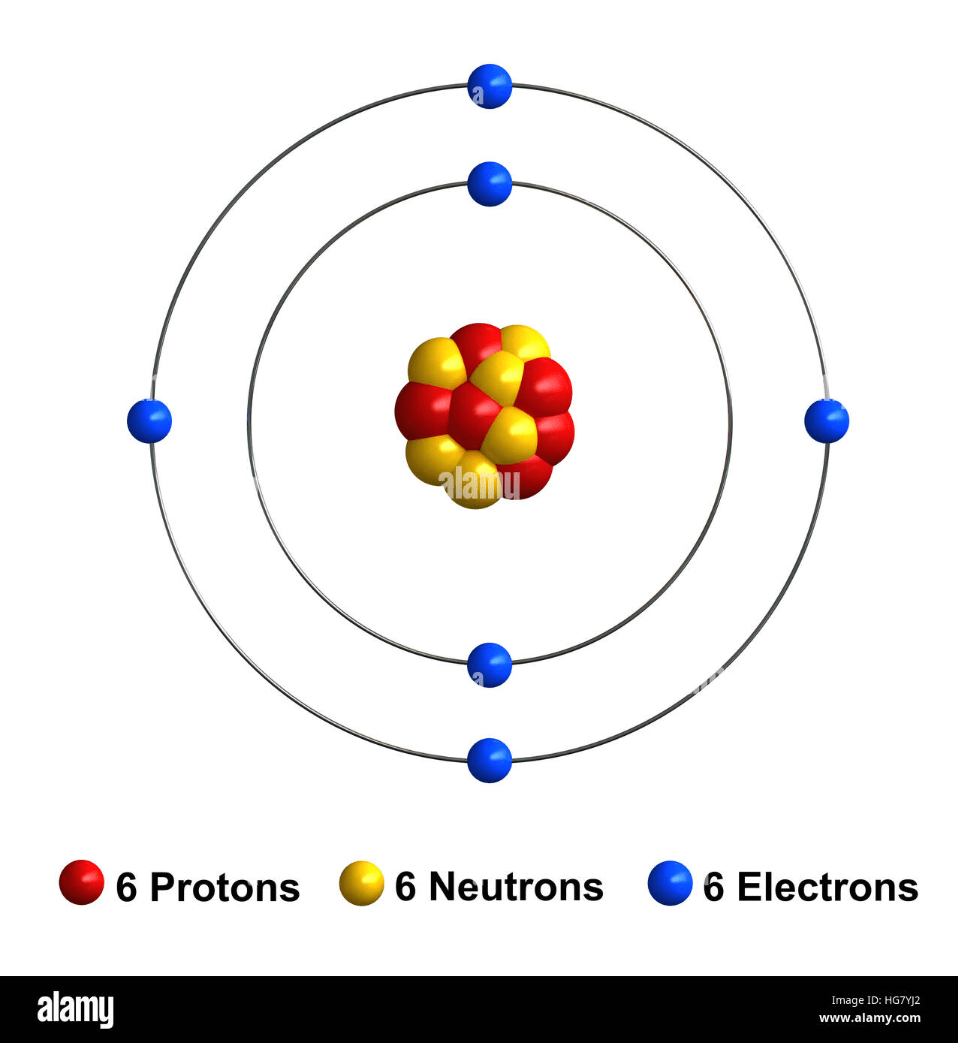
4 electrons
How many protons, electrons, and neutrons in a Zinc atom?

30 protons
30 electrons
35 neutrons
What wild animal attacked Ms. Reed's nephew in their swimming pool?
What is a fox.
The word "atom" comes from the Ancient Greek ἄτομος (átomos), which means..... what?
Uncuttable
Indivisible
What does "periodic" mean, and how does it describe the Table of the Elements?
Periodic describes something that happens at regular intervals, creating a pattern. Elements in the Periodic Table form patterns in their order, period, and group.
This is an oxygen atom. How many bonds can it make with other atoms?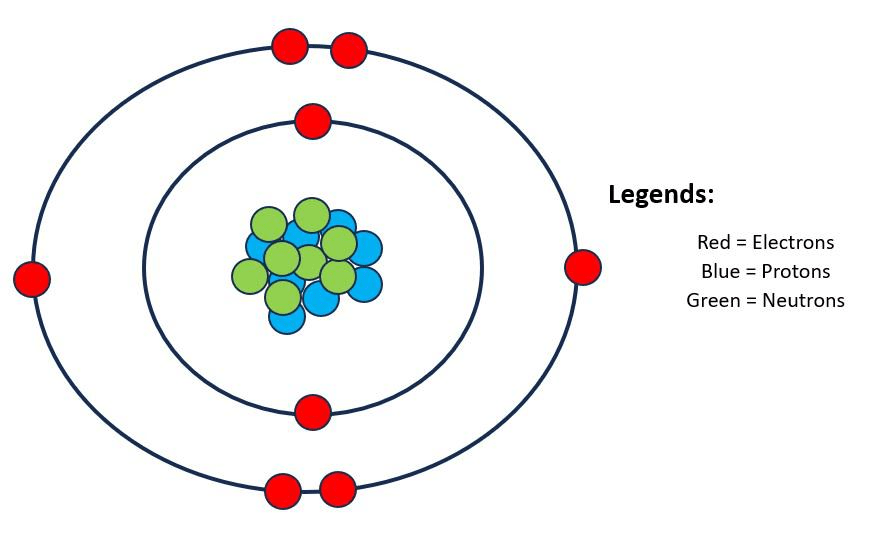
2 bonds.
(It needs 2 more electrons to have a complete outer shell of 8, so it can make 2 bonds)
What element is this?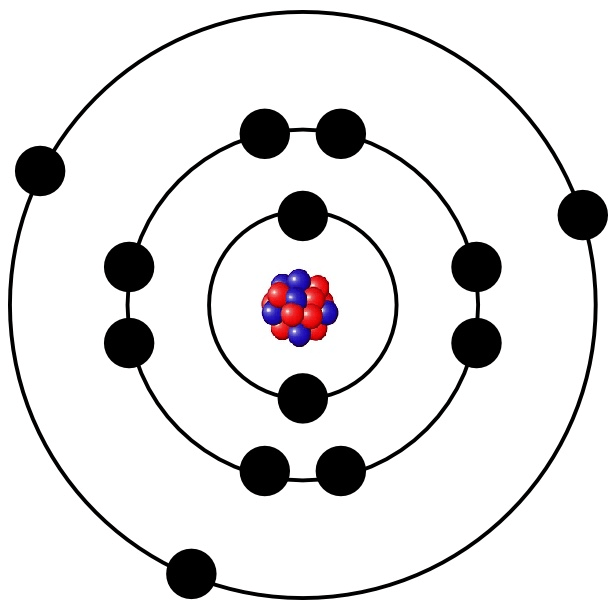
Aluminum
What are Ms. Reed's 2 dogs names?
What are Charlie and Bilbo.
What is an example of something NOT made of atoms?
Answers may vary.
Possible answers: Energy, Thoughts, Love, Light, Sound, Friendship, etc
These are atom from three Noble Gases - Helium, Neon, and Argon. Based on what you know about atomic structure, why are they called "noble" gases?
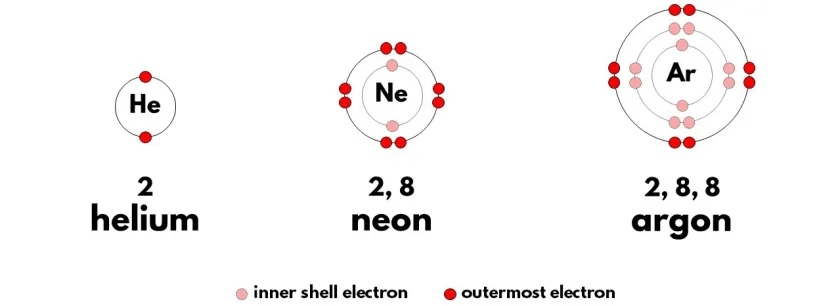
These elements are "Noble" because with full outer shells, they are not reactive and are stable, reliable, safe.
Aka Noble.
Which atom will be more reactive?
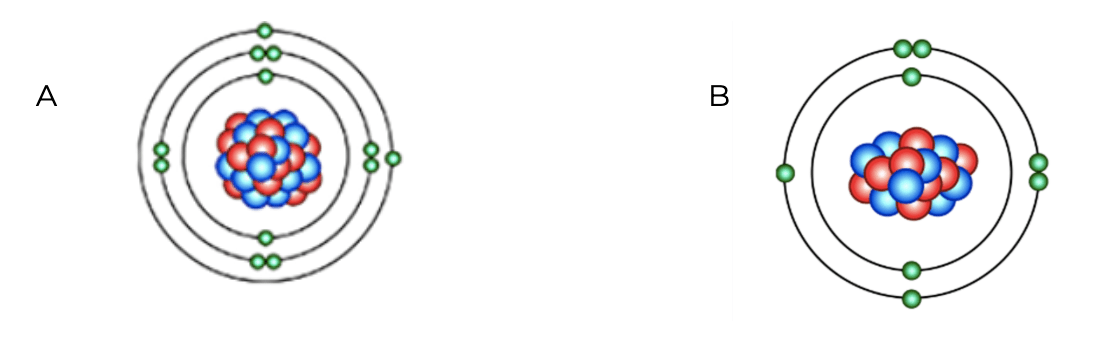
Atom A is more reactive.
This is because A's outer electron shell is almost empty. Atoms whose' outer shells are almost empty or almost full are more reactive.
What is an Ion?
An Ion is an atom that has missing or extra electrons.
An Ion is an atom that has a positive or negative electric charge.
What was Ms. Reed's upbringing like?
What is that she grew up rurally, with no running water and had to use an outhouse.