Atoms are composed on these 3 subatomic particles
What are protons, neutrons, and electrons
How many protons are in an oxygen atom

What is 8?
Two or more different atoms bonded together
What is a compound?
Atomic number of Silicon.
What is 14?
Have no charge and a relative mass of 1.
What are neutrons?
List three properties of metals
Shiny, conduct heat and electricity, malleable, ductile, usually solid at room temperature
A combination of two or more substances
What is a mixture?
The chemical formula for table salt
What is NaCl?
Found outside the nucleus, are negative and have no mass
What are electrons?
The atomic weight is made up of what two subatomic particles. 
What is protons and neutrons?
True or False: Substances in mixtures are chemically bonded together.
What is False?
A mixture of oil and water.
What is a homogeneous mixture?
The center of an atom that contains protons and neutrons
What is the nucleus?
An element that is shiny, brittle and a semi-conductor would be classified as this.
What is a metalloid?
A mixture which has substances that are unevenly distributed.
What is a heterogeneous mixture?
An atom has an atomic mass of 19. If it has 9 protons, how many neutrons will it have.
What is 10?
These move at the speed of light
What are electrons?
The name of the first group on the periodic table
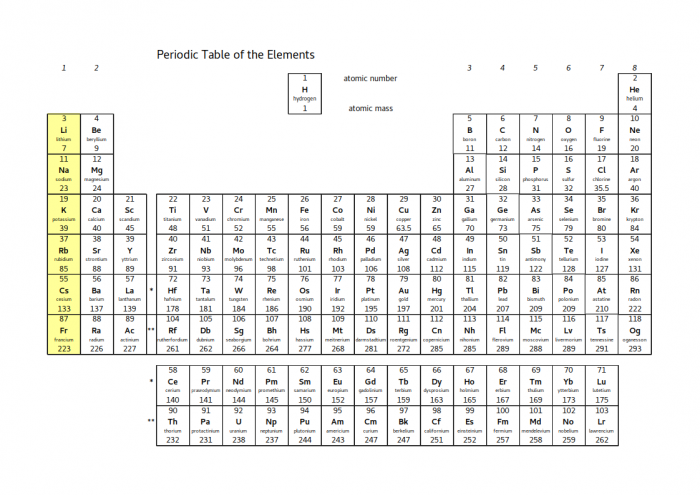
What are alkali metals?
A mixture which insoluble particles that cannot be seen by the naked eye.
What is a colloid?
An atom that has the same number of protons but different number of neutrons.
What is an isotope?