These are pockets within enzymes that specifically binds to substrate and where catalysis happens.
Active sites
These are substances that are tightly or covalently bound to the apoenzymes to make them whole.
Prosthetic groups
The electron transport chain pumps this ion creating the electrochemical gradient.
Hydrogen ion/ Proton
This is the number of electron an oxygen atom can still obtain.
2 electrons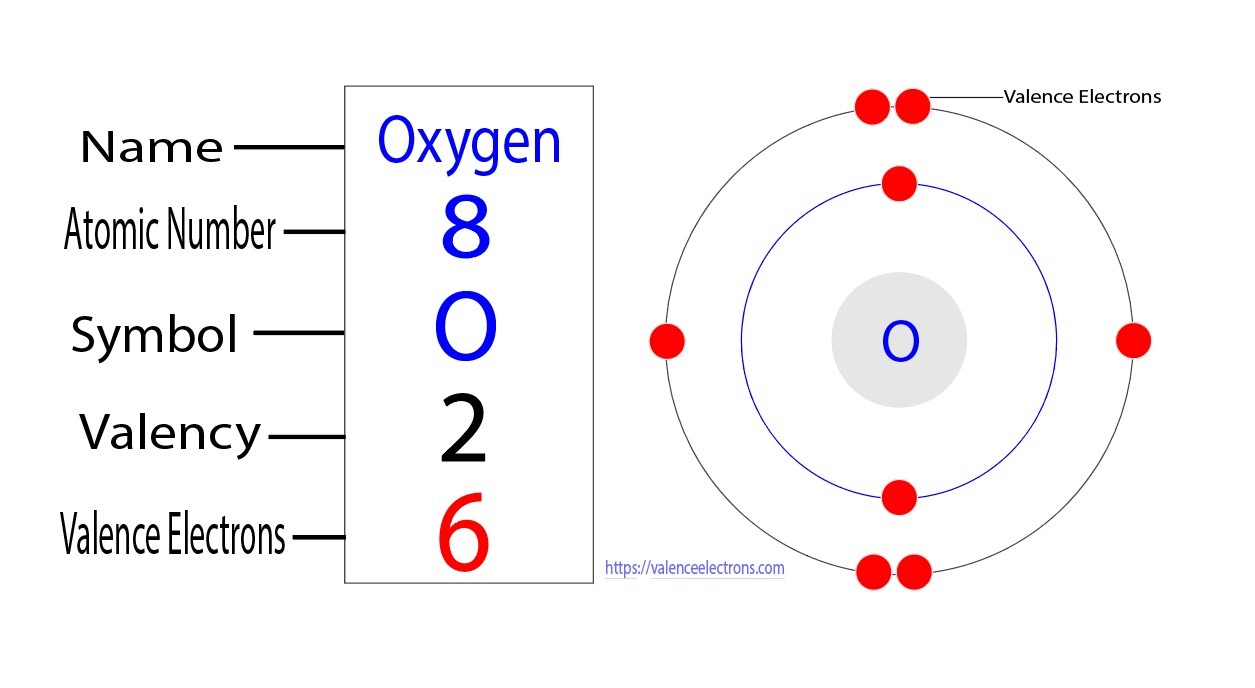
This type of metabolic pathway recovers its substrate at the end of each turn.
Cyclical pathway
This type of long-distance intercellular communication uses ligands which have low affinity to their receptors.
Neurocrine
These are enzymes that cleave giant molecules into smaller substituents replacing single bonds with double bonds.
Lyases
This is the mechanism of action of thiamin.
Transfer of acyl group
This enzyme transfers the electron from NADH to coenzyme Q.
NADH dehydrogenase or NADH-coQ oxidoreductase
This compound is formed when oxygen receive one electron.
Superoxide
This type of reaction is irreversible and generates flux towards the production of the product.
Committed step
This ion is a well known intracellular signal that results into smooth muscle contraction and exocytosis.
Calcium
This parameter activates enzyme by increasing the kinetic energy of the molecules but inactivates them oermanently by denaturation.
Temperature
This enzyme complex utilize five co-enzymes to convert a 3-carbon molecule to 2-carbon molecule.
Pyruvate dehydrogenase
This is the number of ATP produced with one molecule of NADH
3 or 2.5 ATPs
This enzyme catalyze the formation of hydrogen peroxide and water from an oxygen radical.
Superoxide dismutase
In glycolysis, this nucleotide is a potent non-covalent inhibitor.
ATP
This amino acid is found on the cytoplasmic side of the receptor used by insulin and the receiver of phosphate groups during activation.
Tyrosine
This energy is needed to convert the stable substrate into unstable transition states then to stable products.
Activation energy
This substance inhibit the action of biotin leading to hair brittleness and skin dryness.
Avidin
This mitochondrial product transfers electron and is an activator of the intrinsic cell death.
Cytochrome c
This organelle contains important enzymes that converts hydrogen peroxide into water.
Peroxisomes
This term is used for reactions that replenish the substrates of a pathway.
Anaplerotic reaction
This pathway involving enzyme receptors often leads to cellular growth and proliferation.
MAPK pathway
This is the energy to remove layers of water between the active site and substrates.
Desolvation effects
This condition is caused by the deficiency of tetrahydrobiopterin.
Phenylketonuria
The NADH from glycolysis uses this shuttle system to recover 3 or 2.5 ATPs.
malate-aspartate shunt
This tripeptide molecule is used by the body to give electrons to hydrogen peroxide and stabilize them to water.
Glutathione
This gluconeogenesis enzyme replenishes the citric acid cycle.
Pyruvate carboxylase
This enzyme is the effector protein for GPCRs related to light perception.
Phosphodiesterase