What is the difference in where prokaryotes and eukaryotes store their DNA?
Prokaryotes: DNA is in the "nucleoid" region of the cytosol (doesn't have a nucleus)
Eukaryotes: DNA is stored in the nucleus.

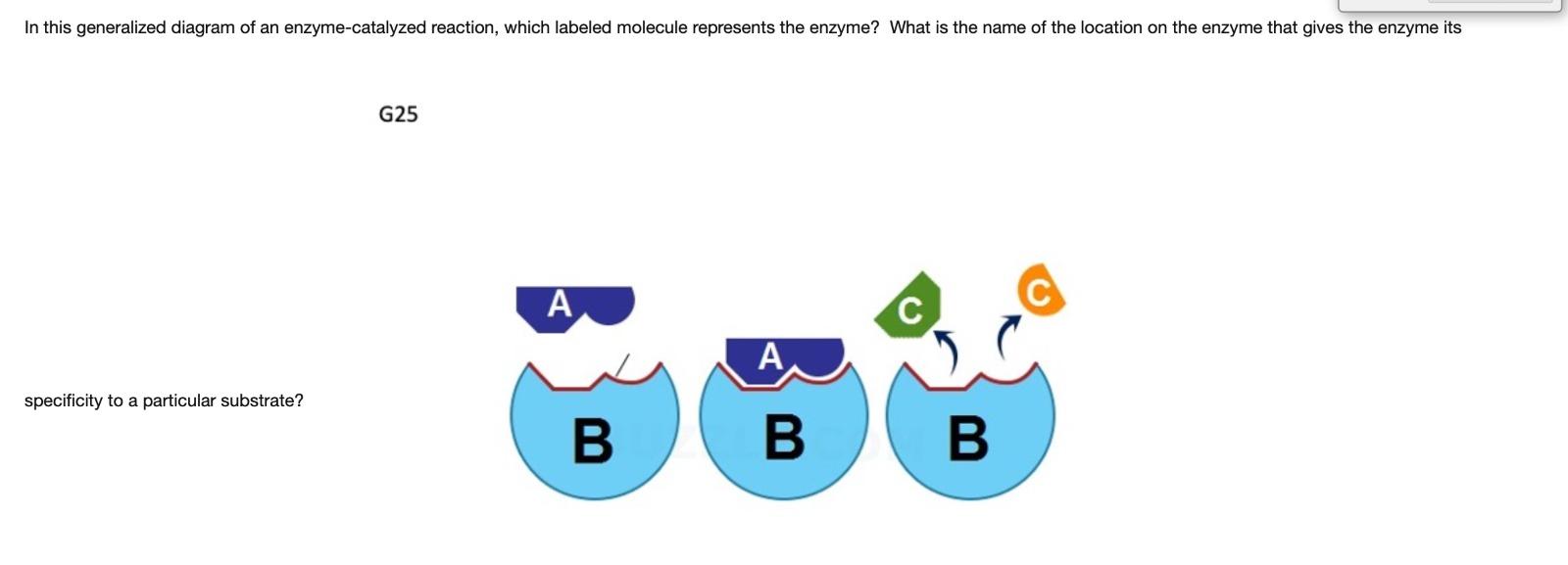
Which letter represents the enzyme? Which letter represents substrate?
Enzyme=B
Substrate=A
List one function of the cytoskeleton.
1. To support the cell and maintain its shape.
2. To help the cell move around (cilia/flagella).
3. To move materials around inside of the cell (like on train tracks).
This is a diagram of glucose. What kind of energy does it contain in its bonds?
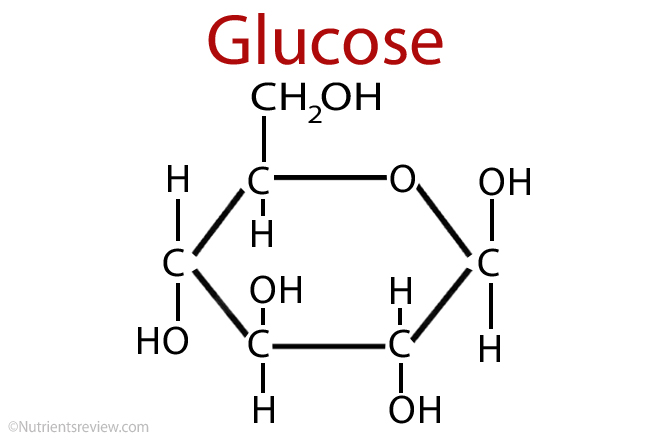
Chemical energy, which is a form of potential energy. Glucose has a high amount of potential energy in its bonds.
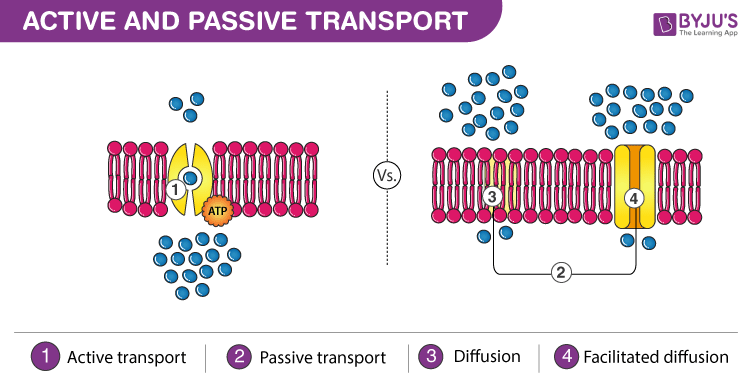
Name at least two differences between passive and active transport.
Passive: movement from high to low concentration, travels down gradient, no energy required. Sometimes uses transport proteins (facilitated diffusion).
Active: movement from low to high concentration, travels up/against gradient, ENERGY REQUIRED. Always uses transport proteins.
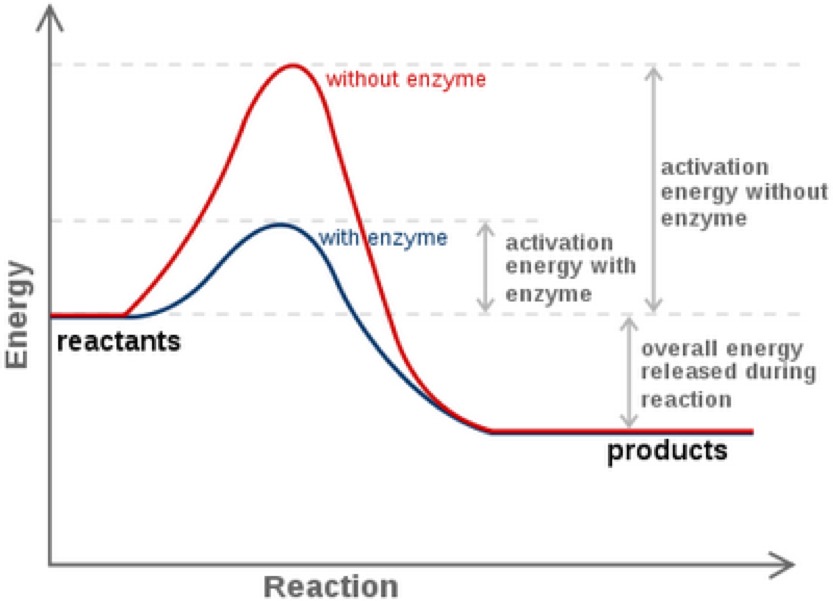
How do enzymes speed up reactions?
They lower the activation energy required for the reaction, allowing the reaction to take place quicker. They do this by drawing in the substrate(s) into the active site and arranging them in a way that is favorable for the reaction to occur.
Pick one of the following types of cell junctions and list its function/job.
1. Anchoring junctions
2. Tight junctions
3. Communicating junctions (gap junctions/plasmodesmata)
1. Anchoring junctions: hold sheets of cells together, but materials can still pass in between the cells.
2. Tight junctions: seals neighboring cells together, so materials cannot pass between cells.
3. Communicating junctions: form passages between cells, so materials can go directly from one cytoplasm to another (gap junctions = animal cells, plasmodesmata = plant cells).
Define "oxidation" and "reduction."
Oxidation: when a molecule loses electrons and hydrogen ions (H+)
Reduction: when a molecule gains electrons and hydrogen ions (H+)
In redox reactions, whenever one molecule is reduced, another is oxidized.
What's the difference between a hypertonic and a hypotonic solution?
Hypotonic solution refers to a solution that has a lower concentration of solute when compared to the cell in the solution. Ex: Solution is 100% water (0 M NaCl), but the cell is 0.1 M NaCl. Solution has less solute than the cell = hypotonic.
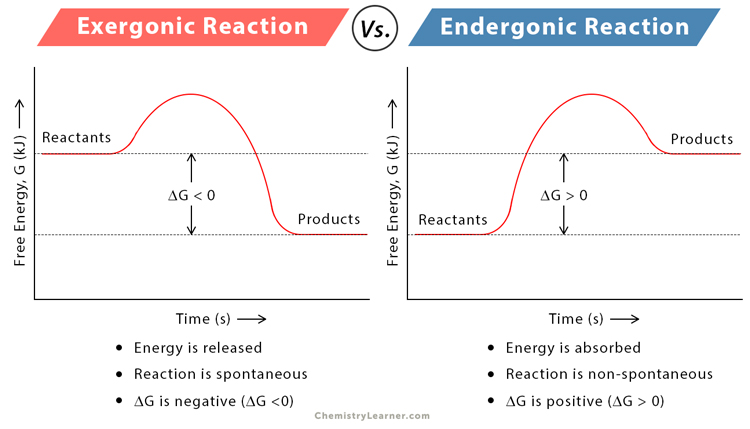
Define "energy coupling."
Energy coupling is when the energy released from an exergonic reaction is used to power an endergonic reaction (that requires energy).
Example: Hydrolysis of ATP (breaking the last phosphate off of ATP) is exergonic and is used to power most endergonic work in the cell.
List the monomers/shape of each of the types of cytoskeleton fibers:
1. Microfilaments
2. Intermediate filaments
3. Microtubules
1. Microfilaments: actin monomers in a double helix shape
2. Intermediate filaments: protein subunits in a cable
3. Microtubules: tubulin dimers in a hollow tube.
List each step of cellular respiration in the correct order, and where does it occur?
1. Glycolysis: cytosol
2. Pyruvate oxidation: Mitochondrial matrix
3. Citric acid cycle (Krebs cycle): Mitochondrial matrix
4. Oxidative phosphorylation (ETC/chemiosmosis): inner membrane of the mitochondria
Rough ER > vesicle > Golgi apparatus > vesicle > cell membrane.
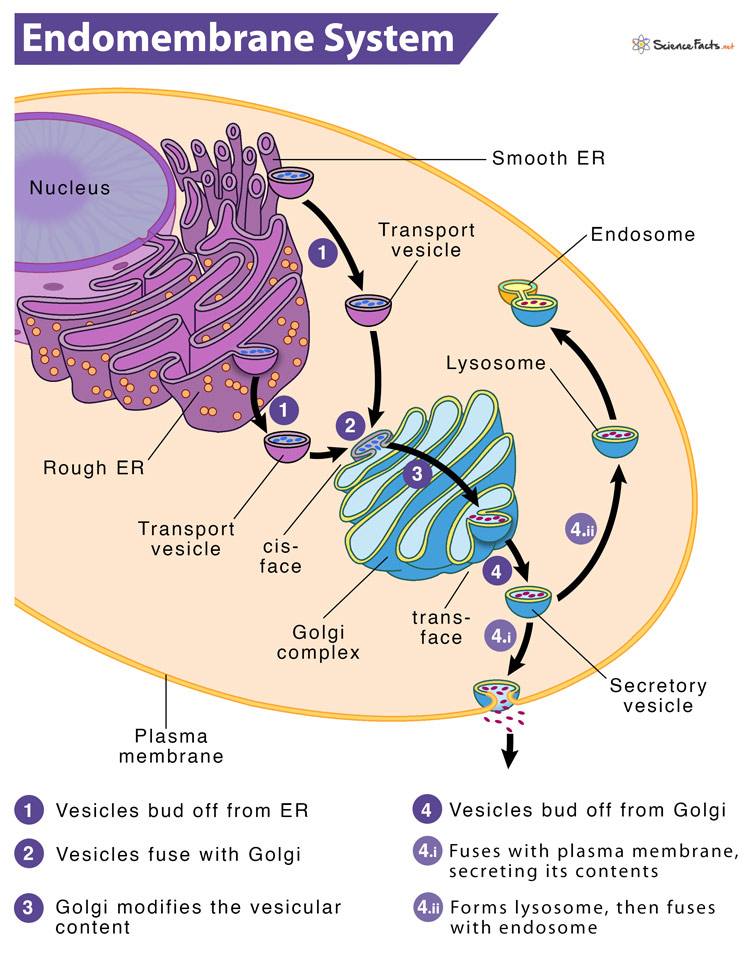
What is the difference between a competitive enzyme inhibitor and a noncompetitive enzyme inhibitor?
Competitive: binds directly to the active site ("competes" with the substrate) and blocks the substrate from binding.
Noncompetitive: binds to a different spot on the enzyme, which changes the shape of the enzyme, and prevents the substrate from binding to the active site (because it no longer matches the shape).

What is the total number of ATP made from 1 glucose molecule by aerobic respiration AND anaerobic respiration?
Aerobic: 32-34 total ATP (2 from glycolysis, 2 from citric acid cycle, 28-30 from oxidative phosphorylation).
Anaerobic: 2 total (2 from glycolysis, none from fermentation).
After the Krebs cycle, but before the ETC, what molecule(s) contain most of the potential energy of the original glucose molecule?
NADH and FADH2 (the electron carriers)
Name 2 ways to increase the fluidity of the plasma membrane.
1. Increase temperature (higher temp=more fluid)
2. Increase number of unsaturated fatty acid tails in the phospholipids (more unsaturated=more fluid)
3. Decrease number of saturated fatty acid tails in the phospholipids.
Make a chart on your whiteboard with "Catabolic" on one side and "Anabolic" on the other. Write these words under the correct category:
Endergonic, Exergonic
Releases energy, Absorbs/Requires energy
Breaks things down, Makes bigger molecules
+change in energy, -change in energy
Catabolic: Exergonic, Releases energy, Breaks things down, -change in energy
Anabolic: Endergonic, Absorbs/requires energy, makes bigger molecules, +change in energy.
Fermentation happens after glycolysis when there is no oxygen present. What is the main purpose of fermentation?
The main purpose is to turn NADH back into NAD+ (because glycolysis needs NAD+ in order to occur). NADH is oxidized back into NAD+ by reducing (giving those electrons to) pyruvate.
Explain the main steps of oxidative phosphorylation (step 4 of cellular respiration).
1. electrons are dropped off at the ETC by NADH/FADH2;
2. energy from e- used to pump H from matrix into intermembrane space;
3. H flow down their gradient through ATP synthase from intermembrane space to matrix, providing the energy to make ATP from ADP;
4. electrons from the ETC and H reduce O2 into H2O (O2 is the final electron acceptor at the end of the ETC).