What is the atomic number?
This number tells you how many protons are in an atom.
What is the pH scale?
This scale measures how acidic or basic a substance is.
What is complementary base pairing?
The specific, hydrogen-bonded pairing of nitrogenous bases in DNA and RNA, where adenine (A) pairs with thymine (T)—or uracil (U) in RNA—and guanine (G) pairs with cytosine (C).
What are amino acids?
The building blocks (monomers) of proteins.
Monomers are small, basic molecular building blocks. 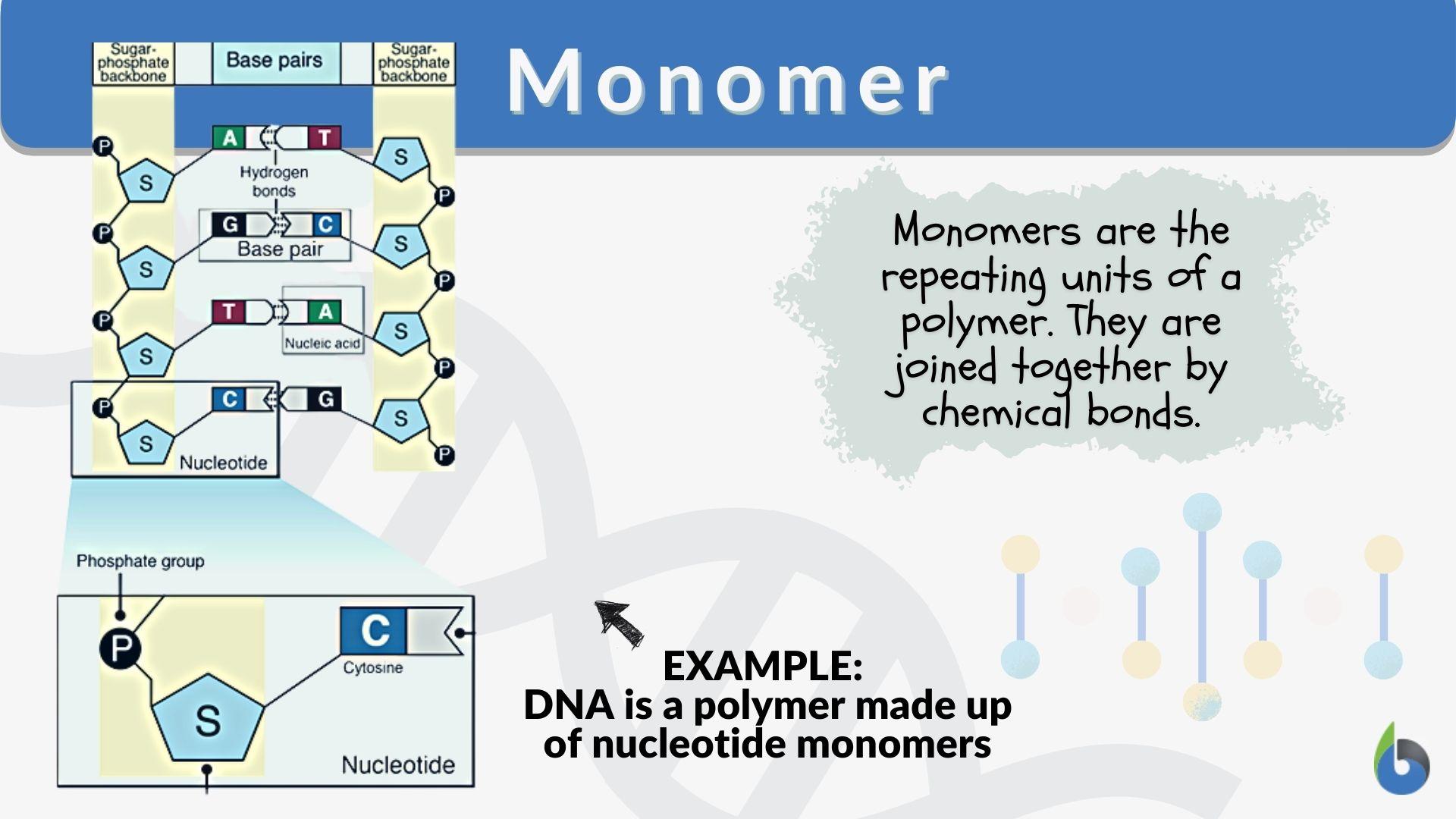
What are reactants?
These are the starting substances in a chemical reaction.
What is a covalent bond?
This bond forms when electrons are shared between atoms.
What is 7 on the scale?
On the pH scale, this number is considered neutral.
What are purines?
Adenine and Guanine belong to this structural group.
What is primary structure?
The sequence of amino acids in a protein is called this level of structure.
What are products?
These are formed at the end of a chemical reaction. 
What is an ionic bond?
This bond forms when electrons are transferred between atoms.
What is the solute?
In a solution, this substance is being dissolved.
What are pyrimidines?
Cytosine, Thymine, and Uracil belong to this structural group.
What is denatured?
When a protein loses its shape due to heat or pH change, it becomes this.
What is a polypeptide?
A long chain of amino acids is called this.
What is neutral charge?
An atom with equal protons and electrons has this type of charge.
What is ice floating?
Water is less dense as a solid than a liquid, which explains this phenomenon.
What is the Central Dogma?
This describes the flow of genetic information from DNA to RNA to Protein.
What is the substrate?
This molecule binds to an enzyme at the active site.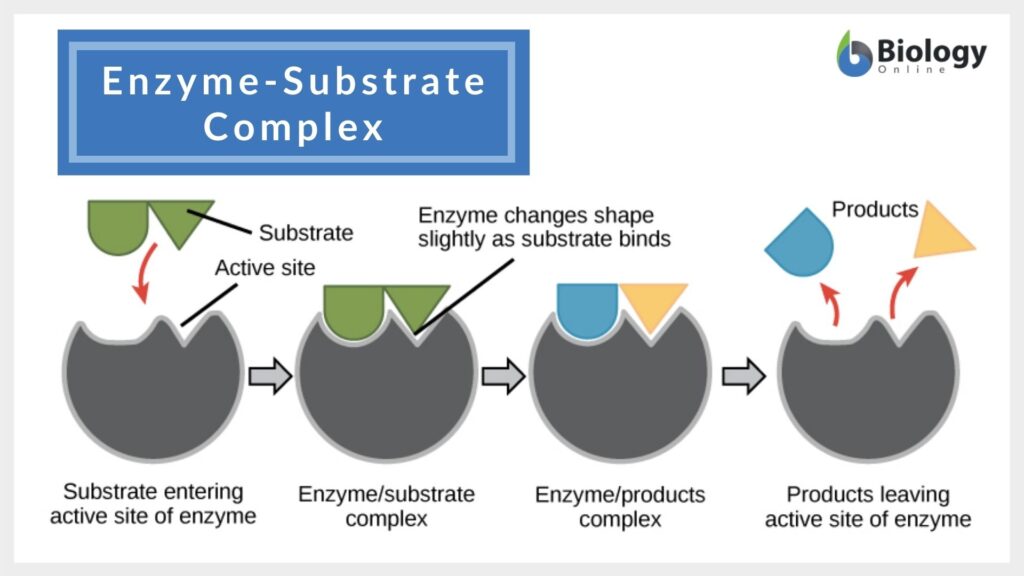
What are oligopeptides?
Short chains of amino acids are called this.
What are orbitals (or electron shells)?
These regions around the nucleus describe where electrons are most likely found.
What are buffers?
These substances resist changes in pH.
What is DNA?
These molecules make up chromosomes along with proteins.
What is sickle cell disease?
A single amino acid substitution in hemoglobin alters folding and changes the overall shape of red blood cells.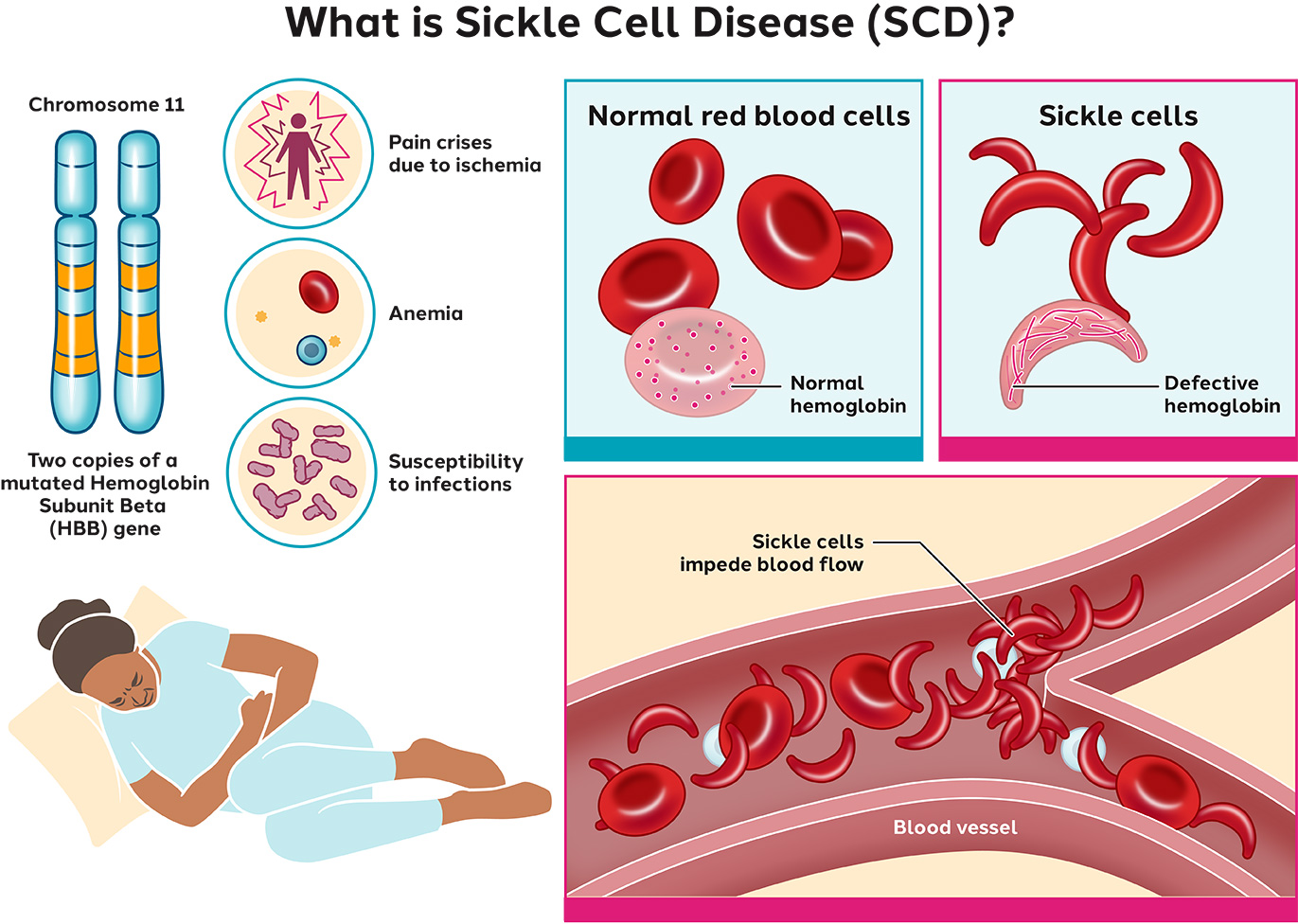
What are allele frequencies?
Natural selection causes changes in this over time within a population.