What is Cellulose?
What is Hydrophobic?
Amino acids are made up of three functional groups; A carboxyl group, an R group, and this group.
What is an Amine Group?
This Nitrogenous Base is found only in RNA
What is Uracil?

This is the name for a mean protein.
What is an A-Mean-o acid?
This is the more common name for Amylose, which is broken down by humans for energy.
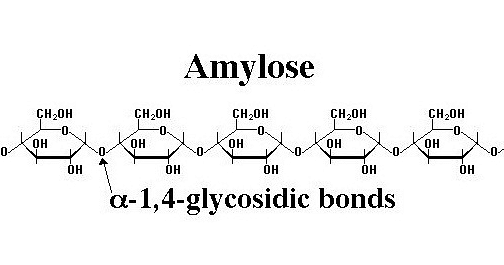
What is Starch?
When a fatty acid contains a Carbon-Carbon double bond, it is called this, which means it is usually in liquid form.
What is unsaturated?
The enzyme lactase's substrate is this polysaccharide.
What is lactose?

This form of RNA is the building material for ribosomes.
What is rRNA?
The given string of Nitrogenous Bases will correspond to this many Amino Acids.
UAGGGCUUUAAG
What is Four?
Deoxyribose and Ribose are examples of monosaccharides with this many carbons.

What are 5 Carbons?

This steroid fat (lipid) is usually found in greater quantities in males.
What is Testosterone?

This protein, found in the blood, contains 3032 carbons, 4816 Hydrogens, and 4 Irons, as well as Nitrogen and Sulfur.
What is hemoglobin?
This molecule, used to store and transport useable energy following cellular Respiration, is a nucleotide.
What is ATP?
Dehydration synthesis is the general term for the reaction that binds together two of these.

What are monomers?
What is a Glycosidic bond?
The head of a phospholipid includes this group, which makes it polar.
What is a Phosphate Group?

These forces are the cause of the folding seen in tertiary structure
What are Hydrophobic/Hydrophilic Forces?
The Nucleic Acid shown below has this structure.
What is Pyrimidine?
Vitamins A, D, E, and K are in a category of vitamins which are soluble in this kind of Biomolecule.

What are Lipids?
This suffix, common in sugars, translates roughly from Latin into "Full of"
What is -OSE? (glucOSE, sucrOSE)
Lipids have many more of these high-energy bonds between these two elements than sugars, making them ideal for energy storage.
What are Carbon and Hydrogen?
This diagram of a protein would be described as this. (relates to it's symmetry)
What is Chiral?
This end of the DNA is the end where the phosphate group is above the sugar.
What is 5'
This element, shown below, appears in these two types of biomolecules.
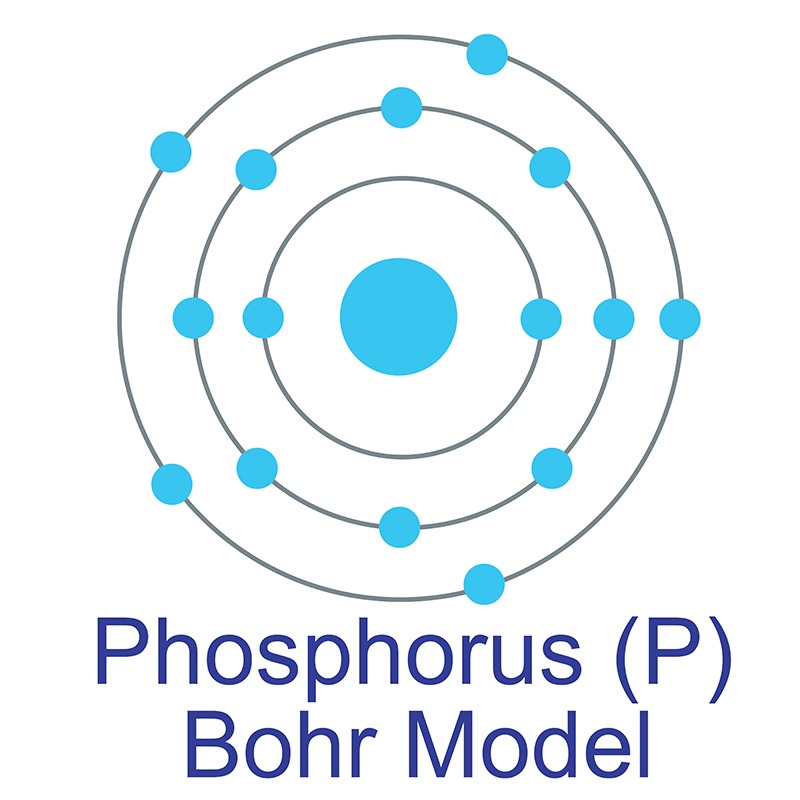
What are Nucleic Acids and Lipids?