Ionic bonding occurs between what 2 types of elements?
metals and non-metals
Covalent bonding occurs between what 2 types of elements?
non-metals and non-metals
What 3 letters do ionic compounds end with?
-IDE
What 3 letters do covalent compounds usually end with?
-IDE
What is special about polyatomic ions?
Covalent compound with a charge
Which electrons are involved in ionic bonding?
valence electrons
Which electrons are involved in covalent bonding?
valence electrons
Name the following compound: LiCl
lithium chloride
What is the prefix for 4 atoms?
tetra-
What is the name of this polyatomic ion (MnO4)?
Permanganate
The criss-cross method is used for what?
writing the formulas for ionic compounds
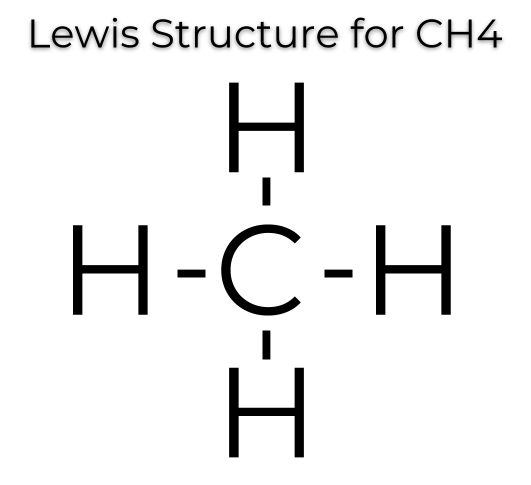
What is the Lewis Structure for CH4?
CH4, carbon tetrahydride
TRUE OR FALSE: The name of CaCl2 is calcium dichloride.
FALSE. The real name is calcium chloride.
Name the following compound: N2O2
dinitrogen dioxide
Name the compound: Li(NO3)
lithium nitrate
Electrons are doing what in ionic bonding?
Electrons are lost or gained
Anion steals electrons away from cations and cations gladly let them.
What is a chemical formula? (you can give an example)
a formula that gives you the atoms and the number of atoms for each element
Ex. H2O or CaCl2
Name the following compound: HgO
mercury (II) oxide
Name the following compound: P2O4
diphosphorus tetroxide
Chemical formula for potassium hydroxide
KOH
What type of ionic compound has only 2 different atoms bonded together?
binary ionic compound
What is the lewis structure of N2?
N triple bonded to N with a lone pair each
What is the name of CuCl2?
copper (II) chloride
what is the chemical formula for tetraphosphorus decoxide
P4O10
chemical formula for potassium phosphate
K3(PO4)