Number of bonds typically formed by Nitrogen
What is 3?
This functional group has the generic formula R-OH
What is alcohol?
Hybridization of the carbon atom in CH4
What is sp3?
According to the VSEPR theory the shape of H2O is this
What is bent or angular?
The original number of allotropes of carbon
What are 2?
The formal charges for the indicated atoms in the molecule below are:
a) -1
b) +1
c) 0
The bond type in CO2
What is polar covalent?
The functional group with a halogen
What is alkyl halide?
An sp2 hybrid orbital is made up of this
What is 1 s and 2 p orbitals?
In the lewis structure for ammonia, there are how many bonds and unshared pairs
What is 3 bonds and 1 unshared pair?
The 2 original allotropes of carbon
What are diamond and graphite?
This element bears a partial negative charge in the H-F bond.
What is F?
The generic formula for an ether
What is R-O-R'?
The bond angle associated with an sp hybridized carbon
What is 180 degrees?
The shape of the lewis structure for methane gas, CH4
What is tetrahedral?
Diamond and graphite are 2 allotropes of carbon, this is the 3rd elemental form
What is the buckyball or fullerene?
Type of bond formed by head to head overlap of s and/or p orbitals
What is sigma?
CH3COOH is an example
What is carboxylic acid?
This type of bond is formed by the overlap of unhybridized parallel p orbitals
What is pi?
H2O is this kind of molecule because it has an unequal charge distribution
What is polar?
This many carbons comprise a Buckyball
What is 60?
The number of pi bonds CH3C≡CCH2CH═CH2
What is 3?
This type of amine has only 1 Hydrogen attached to the Nitrogen atom
What is secondary?
The hybridization of the Oxygen in the molecule shown below.
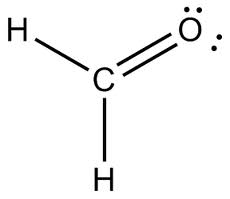
What is sp2?
This is the lewis structure for acetylene, C2H2
What is H - C≡C - H ?
This is the hybridization of diamond which is the hardest known substance on the Mohr's hardness scale
What is sp3?