What are the 4 major macromolecules?
lipids, proteins, carbohydrates and nucleic acids?
The reaction that breaks polymers into monomers by adding water
Hydrolisis
Identify this macromolecule:
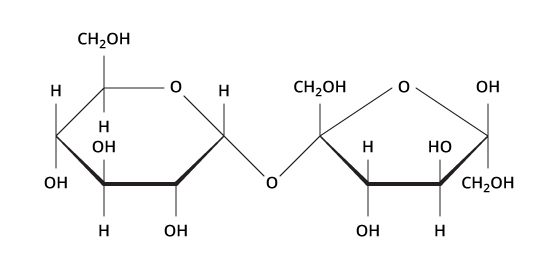
Sucrose (poly)
What is osmosis?
The net movement of water across a selectively permeable membrane following the concentration gradient
What are endo and exocytosis?
Endo: bring things in
Exo: take things out
What is an atom made up of?
In central nucleus: protons, neutrons,
Orbiting clouds around nucleus: (electron shell) electrons
What is thermodynamics and what are it's 1st and 2nd laws?
The study of energy and its transformations
1: Energy cannot be created or destroyed, only transformed (conservation of energy law)
2: When energy ischanged from one form to another, there is a loss of energy that is available to do work. (entropy law)
What is energy and what are its two types?
Energy: the ability to do work or bring about
a change
Kinetic energy:
Energy of motion
Water going over a waterfall
Mechanical
Potential energy
Stored energy
Chemical energy
The food we eat
What are the characteristics of life?
Homeostasis, adaptation/evolution, organized, requires materials & energy, responds to stimuli, reproduces & develops
What is natural selection?
Mechanism of evolution where organisms better adapted to their environment tend to survive and produce more offspring
Charles Darwin
A fatty acid with no carbon–carbon double bonds
saturated fatty acid
A reaction that removes a molecule of water as two molecules become bonded together.
Dehydration synthesis
Identify this macromolecule:

Glucose (mono)
Movement of molecules from high to low concentration without energy input
Passive transport
Describe a hypertonic, hypotonic, and ionic solution
Hypertonic: Solution has more salt than cell. Water leaves cell
Hypotonic: Solution has less salt than cell. Water enter cells
Ionic: Salt levels between solution and cell are equal. Net movement of water is equal
What is pH and its scale?
pH: measure of hydrogen ion concentration
in a solution
pH scale: 0-14
0 to 6.999... = Acidic
7 = Neutral
7+ to 14 = Basic (or alkaline)
Logarithmic scale: Each unit change in pH represents a 10-fold change in H+ concentration
What are enzymes, inhibitors, and active sites?
E: Biological catalysts that speed up reactions
I: A molecule that binds to an enzyme and decreases or stops its activity
AS: The region of an enzyme where the substrate binds
What are reactants and products?
Reactants participate in a reaction.
Products form as result of a reaction.
What is the scientific method and its steps?
Observation
Hypothesis
Prediction(s) and experiment(s)
Data collection w/statistical analysis
Conclusion
What are independent and dependent variables?
An independent variable is the cause, while a dependent variable is the effect in research/an experiment/etc...
A fatty acid whose hydrocarbon chain contains one or more double bonds.
Unsaturated Fatty Acid.
Name each type of saccharide and the macromolecule they are
Monosaccharides, Disaccharides and Polysaccharides (carbs)
Identify this molecule:

Saturated fatty acid
Transport that requires a membrane protein
Facilitated transport
What are crenation, plasmolysis, and lysis?
Crenation: Shrinking of animal cells in a hypertonic solution
Plasmolysis: Shrinking of cytoplasm/other organelles from the plant cell wall
Lysis: Rupturing of a cell in a hypotonic solution
Describe acidic, basic, and neutral, and give at least one example for each one
Acidic (pH 0–6.9): High concentration of H+ ions. Examples include lemon juice, vinegar, and stomach acid.
Basic (pH 7.1–14): Low concentration of H+ ions (or high(er) hydroxide OH-). Also known as alkaline; examples include soap, baking soda, and ammonia.
Neutral (pH 7.0): Balanced and H+ and OH- ions. Pure water
What are endo and exergonic reactions?
Exergonic Reactions: Products have less
free energy than reactants (release
energy). These occur spontaneously.
Endergonic Reactions: Products have more free energy than reactants (require energy input). These are nonspontaneous.
What is photosynthesis?
Biological process by which plants, algae, and certain bacteria convert light energy (usually from the sun) into chemical energy in the form of sugar (glucose)
Name the taxonomic hierarchy
Domain, Kingdom, Phylum, Class, Order, Family, Genus, and Species.
Prokaryote vs Eukaryote?
Proks: No nucleus, no membrane-bound organelles, always unicellular, usually smaller
Euks: Has nucleus, has membrane-bound organelles, usually multicellular, usually bigger
Give an example of a disaccharide
What is sucrose or lactose or...?
What are the monomers for each macromolecule?
Carbohydrates: The monomers are simple sugars, also known as monosaccharides. An example is glucose.
Proteins: The monomers are amino acids
Nucleic Acids: The monomers are nucleotides.
Lipids: Fatty acid and gycerol
Identify this macromolecule:
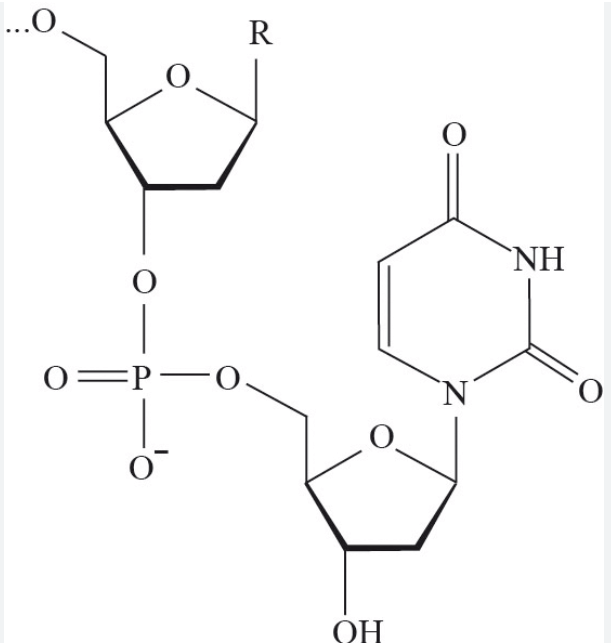
Nucleotide (mono)
Movement of substances against their concentration gradient using energy (ATP)
Active transport
What is turgidity?
Plant cell swollen, distended, or firm, primarily caused by high internal fluid pressure pushing the plasma membrane against the cell wall.
Turgor pressure
Describe neutrons, protons, and electrons
Protons:Positively charged particles found in the nucleus. The number of protons determines the atomic number and identifies the element. They have a mass of approximately 1 atomic mass unit (amu).
Neutrons: Neutral particles with no charge, located in the nucleus with protons. They have a mass of approximately 1 amu. Variations in neutron number create different isotopes of an element.
Electrons: Negatively charged particles found in orbitals surrounding the nucleus. They have a very small mass, approximately 0.0005 amu.
What is ATP?
ATP: Adenosine triphosphate (nucleotide) high-energy compound used as energy in cells. Generated from ADP (extra P).
Coupled reactions
Energy released by an exergonic reaction (or reactions) is
captured in ATP.
ATP is then used to drive an endergonic reaction.
What is cellular respiration?
Metabolic process by which living cells convert nutrients, primarily glucose, into usable energy in the form of adenosine triphosphate (ATP)
Why are cells generally small?
As a cell increases in size, volume increases faster than surface area, limiting efficient exchange across the membrane
What elements are 95% of biological life made out of?
CHNOPS: Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur
The sugar difference between DNA and RNA
deoxyribose vs. ribose
Name the polymers for each macromolecule
Carbohydrates: Polymers are polysaccharides, starch, glycogen, cellulose...
Proteins: Polymers are polypeptides made of amino acid monomers.
Nucleic Acids: Polymers are polynucleotides, DNA or RNA, made of nucleotide monomers.
Lipids: Funky guys! These are not strictly polymers because they aren't composed of long, repeating chains of monomers. Fatty acid/gylcerol chain(s)
Identify this macromolecule:

Polypeptide
What is Bulk transport?
Movement of large materials using vesicles
Name the mechanisms used to help molecules through the cell membrane in facilitated, active, and bulk transport
Facilitated: Channel and carrier proteins
Active: Protein pumps and sodium-potassium pump
Bulk: Endo and exocytosis
Describe the difference between ionic and covalent bonds
Ionic bonds involve the transfer of electrons between metals and non-metals, while covalent bonds involve sharing electron pairs between non-metal atoms
How do living systems comply with the Second Law of Thermodynamics?
Organisms increase entropy in the surroundings by releasing heat during metabolism
Come up to the board and draw + label a plant cell!
:D
Come up to the board and draw + label an animal cell!
:D
What type of intermolecular force gives water its cohesion and high specific heat?
Hydrogen bonding