particles in this state vibrate about a fixed position
What is solid?
the total kinetic energy of a substance
What is thermal energy
Who's boat held the most pennies?
Ella's
the formula for Boyle's Law
What is P1V1 = P2V2
Gas law demonstrated by the balloon in the liquid nitrogen.
What is Charles Law?
particles in this state are easily compressed
What is gas?
a state of matter where atoms and molecules can move past each other
What is fluid?
buoyant force on an object is equal to...
What is "the weight of the water displaced by the object"
Boyles Law states that as the volume of a gas increases it's pressure ______________.
decreases
The liquid tested with the lowest viscosity
What is water?
the most common state of matter in the universe
What is plasma?
51 g = ____ N
0.5 N?
the law that relates the volume and temperature of a gas
What is Charles Law?
gas law demonstrated using the syringe and marshmallows
What is Boyle's Law?
has a definite volume but an indefinite shape
What is liquid
the units for buoyant force
What are newtons?
Gay-Lussacs law says that the ______________ of a gas increases as the temperature __________________ .
What is pressure, increases
Change of state that caused the balloon to inflate when you put dry ice in it.
What is sublimation?
State(s) of matter where particles are free to move past each other.
What are liquids and gases?
the upward force exerted on an object immersed in or floating on a fluid
What is buoyant force?
the force that opposes buoyant force
What is gravity?
A gas occupies 2.5 L at a pressure of 3.0 atm. What will be the volume at a pressure of 3.5 atm?
2.9 L?
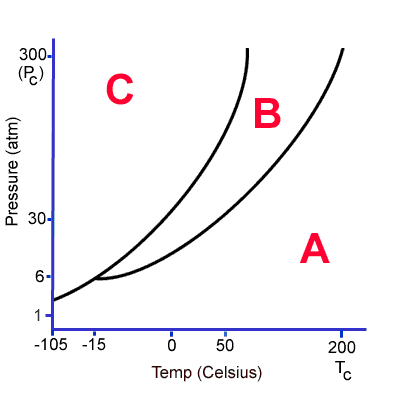
Name boiling point for this substance.
What is 30 atm, 50 degrees C? Other correct answers as well.