The weakest intermolecular force
What are London Dispersion Forces?
This is the definition of molality
What is moles of solute per kilogram solvent?
This is the process when water vapor turns to frost on a car windshield.
What is deposition?
This type of molecule, when incorporated into a mixture of immiscible liquids, prevents/inhibits the coalescence of the dispersed liquid
What is an emulsifier?
Raising the temperature does this to the reaction rate
What is raise/increase the reaction rate?
The intermolecular forces found in butanol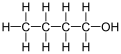
What are London Dispersion, Dipole-dipole, and Hydrogen bonding?
The units for mole fraction xA
What is dimensionless?
xA = moles A/total moles
This type of solid exhibits a well-defined and repeating three-dimensional pattern of particles held together by strong covalent bonds.
What is a covalent network solid?
Adding sucrose to water does this to the boiling point
What is raise/elevate the boiling point?
This is the impact of the use of a catalyst.
What is lowering the activation energy / increasing the rate of reaction?
The molecule with a higher boiling point
Ethane 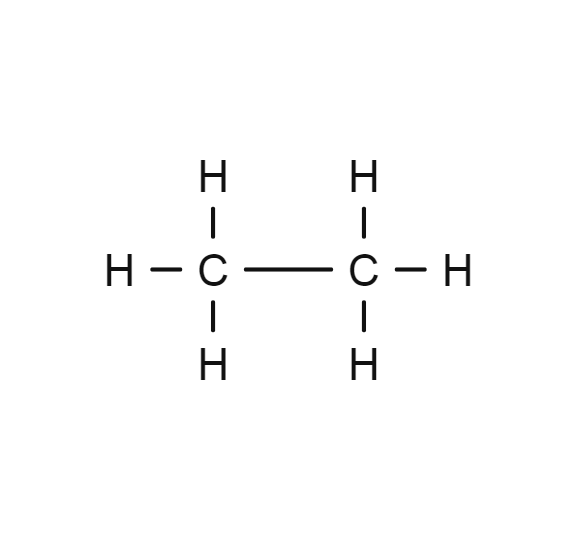
Hexane 
What is hexane?
This is the osmotic pressure of an aqueous solution containing 0.02 M solute at 25 C.
R = 0.0821 (L * atm) / (mol *K)
What is 0.5 atm?
The processes occurring at point A
What are melting/freezing?
The continuous phase in smoke, a colloid, is this.
What is air?
This is the reaction rate with respect to B when the reaction A + B → C has the following rate equation: rate = k[A][B]2
What is second order?
The substance with the lower vapor pressure
CHF3
OR
CF4
What is CHF3?
This is the freezing point of a 0.325 m solution of a nonvolatile nonelectrolyte solute in carbon tetrachloride.
Freezing Point pure solvent -23 C
Kf = 29.8 K kg mol-1
What is -33 C?
This amount of heat required to convert 50.0 g ice at -25.0 °C into water vapor at 105. °C
Specific heat capacity of ice = 2.09 J/g°C
Heat of fusion = 334 J/g
Specific heat capacity of water= 4.18 J/g°C
Heat of vaporization = 2260 J/g
Specific heat capacity of steam = 2.01 J/g°C
What is 153 kJ?
You add 5.67 g of glucose (C6H12O6) to 25.2 g of water at 25 C. The vapor pressure of pure water is 23.8 mm Hg at 25 C. This is the vapor pressure of the solution.
What is 23.3 mm Hg?
The decomposition of NO2 is second order in NO2 with k = 0.543 M-1s-1. If the initial concentration of NO2 is 0.0500 M, this is the concentration after 375 seconds.
What is 4.47 x 10 -3 M?