What is the order of the reaction given a plot of 1/[A] vs time gives a straight line?
Second order
What is the pH of a saturated solution of Ni(OH)2? Ksp = 2.0 x 10-15.
pH = 9.20
Which substance has greater entropy: O2(g) or O3(g)?
O3(g)
For a reaction in a voltaic cell, both entropy and enthalpy are positive. How will Eocell change if we increase the temperature?
It will increase
True or False?
1. Beta particle emission produces a nucleus with one additional neutron and one additional proton.
2. No stable nuclides are known above atomic number 83.
3. In electron capture, a proton is converted to a neutron through the capture of an inner orbital electron.
1. F
2. T
3. T
As the number of solute particles in a given volume of solution increases, what effect does it have on the freezing point and vapor pressure?
Both freezing point and vapor pressure will decrease.
Given Ag+(aq) + 2NH3(aq) <--> Ag(NH3)2+, how will the solubility of an aqueous solution of AgBr(s) change if we add NH3?
The solubility will increase by removing Ag+.
If a reaction is nonspontaneous at 2200K, but spontaneous at room temperature, which statement is false?
1. The change in enthalpy is the main driving force of the reaction.
2. Both entropy and enthalpy are negative for this reaction.
3. Gibbs Free Energy becomes zero at a temperature between 300 and 2200 K.
4. The change in entropy is the main driving force of the reaction.
4. The change in entropy is the main driving force of the reaction.
Given the following standard reduction potentials:
Cl₂ + 2e⁻ → 2Cl⁻ E° = 1.36 V
Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O E° = 1.33 V
Determine the stronger oxidizing agent.
Cl2
Which reaction corresponds to the fusion of deuterium and tritium (2 neutrons and a proton), which forms a single neutron and some other product?
a) 31H + 11n --> 2 21H
b) 31H + 21H --> 42He + 10n
c) 31H + 21H --> 2 11H + 3 10n
d) 21H + 11H --> 31H + 01n
b) 31H + 21H --> 42He + 10n
A 15.7% oil of cinnamaldehyde (C9H8O) in chloroform (Cl3CH) solution is prepared. At 35.0oC, the density of the solution is 1.483 g/mL and the vapor pressure of the solvent, Cl3CH, is 295 torr. The normal freezing point for Cl3CH is -63.5oC, Kf = 4.68oC/m.
What is the vapor pressure and the freezing point of the solution?
Vapor pressure = 252.4 torr
Freezing point = -70.1oC
Will the solubility of Cd(OH)2 increase or decrease when Ba(OH)2 (strong base) is added to the solution? What is the solubility of Cd(OH)2 in 0.350 M Ba(OH)2? Ksp for Cd(OH)2 is 6.50 x 10-6.
Decrease, 1.3 x 10-5 M
What is the equilibrium constant at 400.0K for the following reaction?
2C(s) + 3H2(g) --> C2H6(g); DeltaH^o=-84.68 kJ DeltaS^o = -173.8 J/K at 298 K.
K = 95
You construct a voltaic cell from the following reduction half-reactions:
ZnO2(s) --> Zn(s) E = -1.473 V
NO3-(aq) -> NO(g) E = 0.96 V
Write the balanced chemical reaction in an acidic solution, and determine the standard cell potential and Gibbs Free Energy.
If you connect a voltmeter and get a reading of 2.245 V, the reaction produced [NO3-] = 0.0250 M, and the partial pressure of NO = 0.0350 atm, what is the pH meter reading?
4H+(aq) + 4NO3-(aq) + 3Zn(s) --> 4NO(g) + 2H2O(l) + 3ZnO2(s)
Ecell = 2.433 V
G = -2817 kJ/mol
pH = 9.38
A radioactive isotope decays by alpha emission, followed by two beta emissions. What is the change in mass number and atomic number of the original isotope?
The mass number decreases by 4, and the atomic number is unchanged.
A 10 mL reaction vessel initially contains 2.60 x 10-2 mol N2 and 1.30 x 10-2 mol H2O. At equilibrium, the concentration of NO in the vessel is 0.412 M. What is the value of the equilibrium constant Kc? Is it product or reactant favored?
Kc = 74.9
Product favored
A beaker contains an aqueous solution of 25 mL of 0.25 M (CH3)2NH2NO3. What type of molecule is in the beaker?
a) Will it react if 15 mL of 0.15 M KOH is added? If so, write the reaction and determine what type of solution is left in the beaker. If no, what happens?
b) What about if we add 15 mL of 0.15 M HCl instead?
acidic salt
a) reacts, makes a buffer: (CH3)2NH2+ + OH- --> (CH3)2NH + H2O
b) no reaction, shifts the equilibrium left, common ion effect
To synthesize a dipeptide, the body couples the following two reactions. Write the overall reaction and determine whether it will be spontaneous.
amino acid A + amino acid B ⇌ dipeptide + 2 H₂O
(ΔG = +35 kJ/mol)
ATP + H₂O ⇌ ADP + Pi
(ΔG = -30 kJ/mol)
The reaction will be spontaneous.
amino acid A + amino acid B + 2ATP <--> dipeptide + 2ADP + 2Pi ( DeltaG = -25 kJ/mol)
A current of 8.0 A is passed through a silver nitrate (AgNO₃) solution. How long, in hours, must this current be applied to plate out 15 g of silver? The molar mass of silver is 107.87 g/mol. Hint: (Ag+ + e- → Ag)
0.466 hours
I-127 is the only stable isotope of iodine. Determine the primary mode of decay for I-131 and write the balanced nuclear reaction.
Decay by beta emission
n/p ≈ 1.47 → Too many neutrons
13153I → 13154Xe + 0-1B
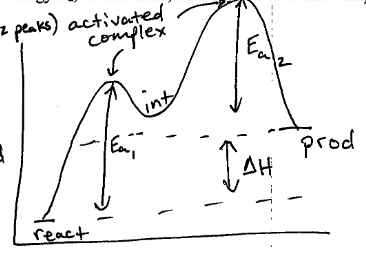
H2O2 + Cl- --> H2O + ClO- fast
H2O2 + ClO- --> Cl- + O2 + H2O slow
Determine the overall reaction, intermediate, catalyst, and the rate law. Draw the energy diagram, label activation energy, intermediate, enthalpy, and activated complex.
H2O2 --> O2 + 2H2O
int = ClO- cat = Cl-
Rate = k([H_2O_2]^2[Cl^-])/([H_2O])
50 mL of a 0.37 M solution of methylamine (CH3NH2, Kb = 4.4 x 10-4) was titrated with 0.52 M HCl. Determine the pH of the solution initially and at the equivalence point.
initially: pH = 12.11
equivalence point: pH = 5.65
The reaction X + 2Y ⇌ Z was run with initial concentrations of [X] = 0.300 M, [Y] = 0.400 M, and [Z] = 0 M. If the equilibrium concentration of Z is 3.2 × 10⁻³ M, determine ΔG for the reaction at 298 K.
ΔG = -6.6 kJ/mol
A voltaic cell is constructed using the following half-reactions:
Zn2++2e− → Zn E∘ = −0.76 V
Cu2++2e− → Cu E∘ = +0.34 V
Calculate the standard cell potential (E°cell) and the cell potential (Ecell) when the concentration of Zn²⁺ ions is 0.10 M, and the concentration of Cu²⁺ ions is 0.50 M.
E°cell = 1.1 V
Ecell = 1.12 V
Gallium-67 has a half-life of 78.3 hrs.
a) Is it above or below the band of stability?
b) What mass ( mug ) is required to generate a radiation source with an activity of 45.1 Ci?
c) How long does it take for Gallium-67 to reach 17.5% of its original amount?
d) Determine the binding energy (J/nucleon) (mp = 1.00728 amu, mn = 1.008665 amu, atomic weight of Gallium-67 = 66.9282049 amu)
a) below
b) 75.5 mug
c) 197 hrs
d) 1.3598 x 10-12 J/nucleon