What are the 4 indicators of a chemical change?
1. Color change
2. Temp change
3. Formation of a gas
4. Formation of a precipitate
What is the mole ratio between sodium iodide and iodine gas in this chemical equation?
2Na + I2 --> 2NaI
2: 1
What are the diatomic elements? & What state of matter are they in their natural state?
H, O, F, I, N, Br, Cl
Gases in their natural state
Sodium chloride was dissolved in an aqueous solution. Identify the solute and solvent.
Sodium chloride is the solute, water is the solvent
What is the pH range of an acid? What is the pH range for a base? What pH is neutral?
Acid: 0-6
Base: 8-14
Neutral: 7
What type of chemical reaction is this? Is it balanced? If not, what are the coefficients that balance this equation?
CaO + H2O --> Ca(OH)2
Synthesis/Combination
Balanced already: 1, 1, 1
How many grams are in one mole of Zinc (Zn)?
65.39 grams
What is Avogadro's number and what does it represent?
6.02X1023
# of atoms/molecules/particles that are in one mole of a substance
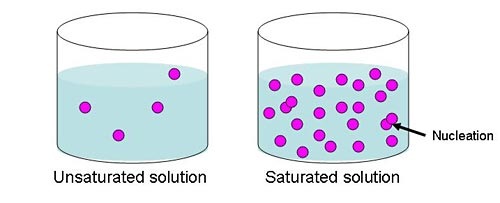
Draw a particle diagram of a saturated solution vs unsaturated solution.
What ions do acids produce in an aqueous solution? What ions do bases produce in an aqueous solution?
Must have the correct charges on ions to be correct!
H+, OH-
Why is the law of conservation of mass important to chemical reactions?
States that mass cannot be created or destroyed, so the same number of atoms in the reactants must be in the products (but they are rearranged).
What is the molar mass of baking soda (sodium bicarbonate) NaHCO3? DON'T FORGET THE UNITS.
84.01 grams/mol
What is the molar mass of glucose (C6H12O6)? DON'T FORGET THE UNITS.
180.16 grams/mol
How does evaporation change the concentration of a saturated solution?
Does not change the concentration. Maximum concentration is maintained.
What is the equivalence point in a Titration and how do we know when it's been reached?
moles acid = moles base
We use an indicator to be able to tell with a color change when the solution has been neutralized.
Predict the products of this reaction:
CH4 + O2 -->
CH4 + 2O2 --> CO2 + 2H2O
2N2 + 5O2 --> 2N2O5
Given this equation, if you had 15 moles of dinitrogen pentoxide, how many grams of oxygen gas would you need?
1200 grams of O2
How many molecules of water are in 300 grams?
1.00X1025 molecules of water
12.24%
Your solution of acetic acid was colorless, but turned pink when 0.2 M sodium hydroxide was added. What indicator was used?
Phenolphthalein
Write the chemical form of this equation, identify the type, and balance:
vanadium + fluorine gas --> vanadium (V) fluoride
2V + 5F2 --> 2VF5
Synthesis
Fe2O3 + CO --> 2Fe + 3CO2
Given the reaction above, how many grams of Fe are produced from 160 grams of iron (III) oxide?
111.96 grams of Fe
C6H12O6 --> 2C2H5OH + 2CO2
Given this reaction, how many grams of carbon dioxide are produced from 180 grams (1 mole) of glucose?
87.94 grams of CO2
What is the molarity of a solution that has 25 grams of sodium chlolride in 500 mL of water.
0.86 M
MB = 0.84 M