The intramolecular attraction happening when the electronegativity difference is 1.7
What is ionic bond?
They molecular formula for sodium and chlorine when bonded?
What is
NaCl
What is the molecular geometry of CO2
Linear
The Electron domain of CO2
What is linear?
Draw the Lewis structure for H2S.
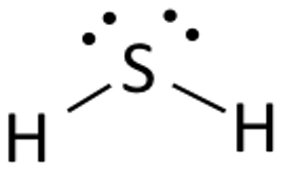
The intramolecular force occuring between hydrogen and oxygen because of its electronegativity difference? 
What is polar covalent bond?
What is BeBr2
What is the Molecular geometry of SiO2
Linear
The Electron Domain of NO3-
What is trigonal planar?
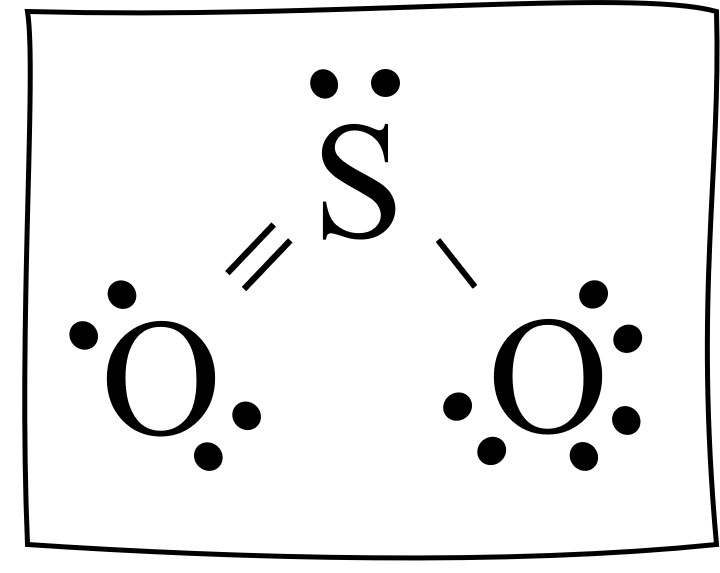
Draw the Lewis structure for SO2.
The type of bond between Cl2
Non-Polar Covalent
The molecular formula between Boron and Sulfer?
B2S3
What is the molecular geometery of NH3
Trigonal Pyrimdal
The Electron domain name of SO2
What is Trigonal Planar
Draw the Lewis structure for GeO2 and tell how many electron domains it has.
What is 2?

The type of bond between Magnesium and Chlorine? 
What is ionic bonding?
The molecular formula between Silicon and Aluminum?
What is the molecular geometery of H2O
Bent
The Electron Domain of OF2
What is Tetrahedral
Draw the Lewis structure for SbCl3 and tell how many bonds and lone pairs there are in it.
What is 3 bonds and 1 lone pair?

The type of bond between Arsneic and Germanium? 
What is non-polar covalent bonding?
the molecular forumla between Indium and Tin?
In4Sn3
What is the Molecular geometry of NO2-1
Bent
The Electron Domain of of SnH3-1
What is tetrahedral
Draw a Lewis Structure for BiF2-1 and tell its number of bonds and lone pairs, and its molecular geometry.
What is 2 bonds, 2 lone pairs, bent