Why do elements form bonds?
To have full valence shells to be stable
A positively charged ion is called a/an _____.
cation
Which element is the MOST electronegative?
Fluorine
Does Li₂O contain ionic or covalent bonds?
ionic
Which group of non-metals does not tend to form bonds due to their full valence shell?
Noble gases
This bond is when two or more atoms share electrons.
covalent bond
A negatively charged ion is called a/an _____.
anion
True or False: ALL elements want eight valence electrons.
False. (ex: hydrogen and helium need only 2, sulfur and phosphorous can hold more than 8, boron is happy with 6)
Does HF contain covalent or ionic bonds?
covalent
What is the name of MgCl₂?
Magnesium chloride
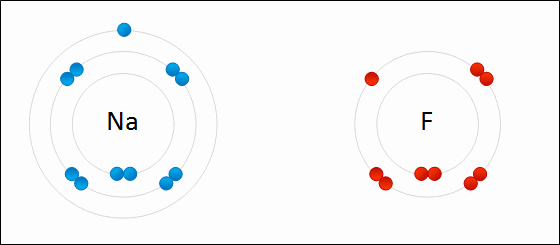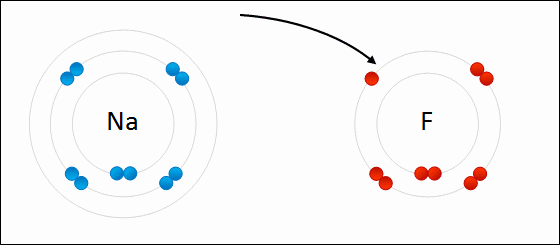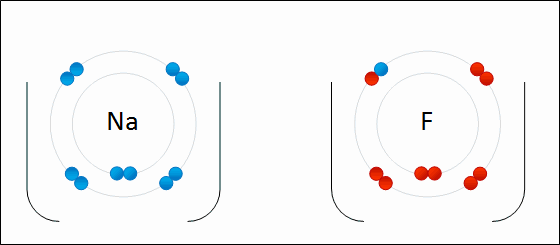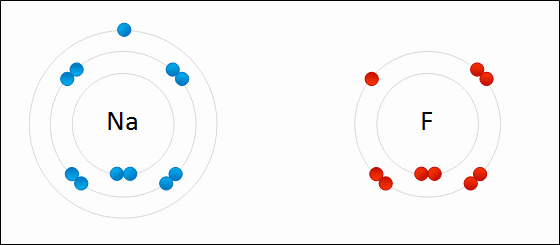
The bond formed when two or more atoms transfer electrons.
ionic bonding
Name the following: Na2S
Sodium Sulfide
This type of covalent bond has an equal sharing of electrons.
Non-polar covalent bond.
Does CO₂ contain single, double, or triple covalent bonds?
double
Ionic bonds form between what types of elements?
Metals and non-metals
If an atom loses an electron, then that atom will become ____ charged.
positively charged
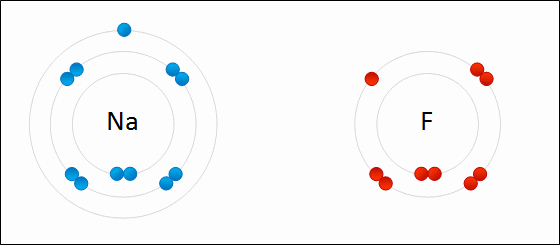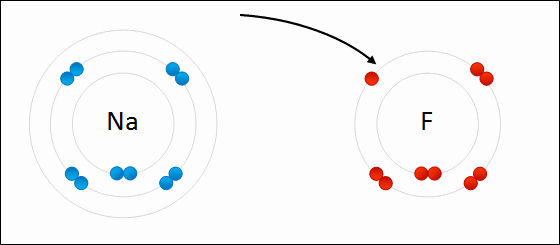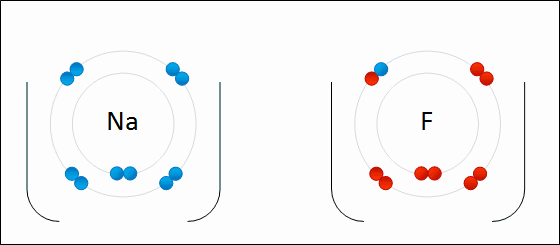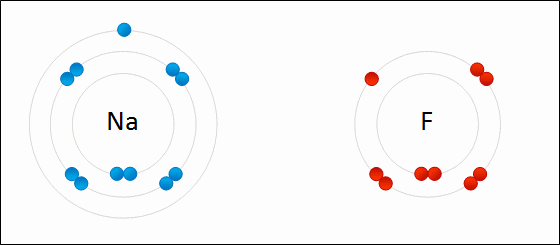
Provide the chemical formula for calcium bromide.
CaBr2
Which periodic trend best explains why non-metals share electrons?
Electronegativity
Would Cl₂ have a nonpolar covalent bond or polar covalent bond?
nonpolar covalent bond
What is the VSEPR shape of CCl4?
Tetrahedral
If an atom gains an electron, then that atom will become ____ charged.
negatively charged

Provide the chemical formula for iron (III) nitrate.
Fe(NO3)3
This type of covalent bond has an unequal sharing of electrons.
Polar covalent bonds
Would HBr have a polar covalent bond or nonpolar covalent bond?
polar covalent bond
What is the name of PCl₅?
Phosphorus Pentachloride
What specific type of electrons are responsible for the reactivities of the elements?
valence electrons
outermost electrons
electrons in outermost shell
Provide the name for Mn(SO4)2
Manganese (IV) Sulfate
Which bond is represented by 2 lines and shares a total of 4 electrons?
Double Bond
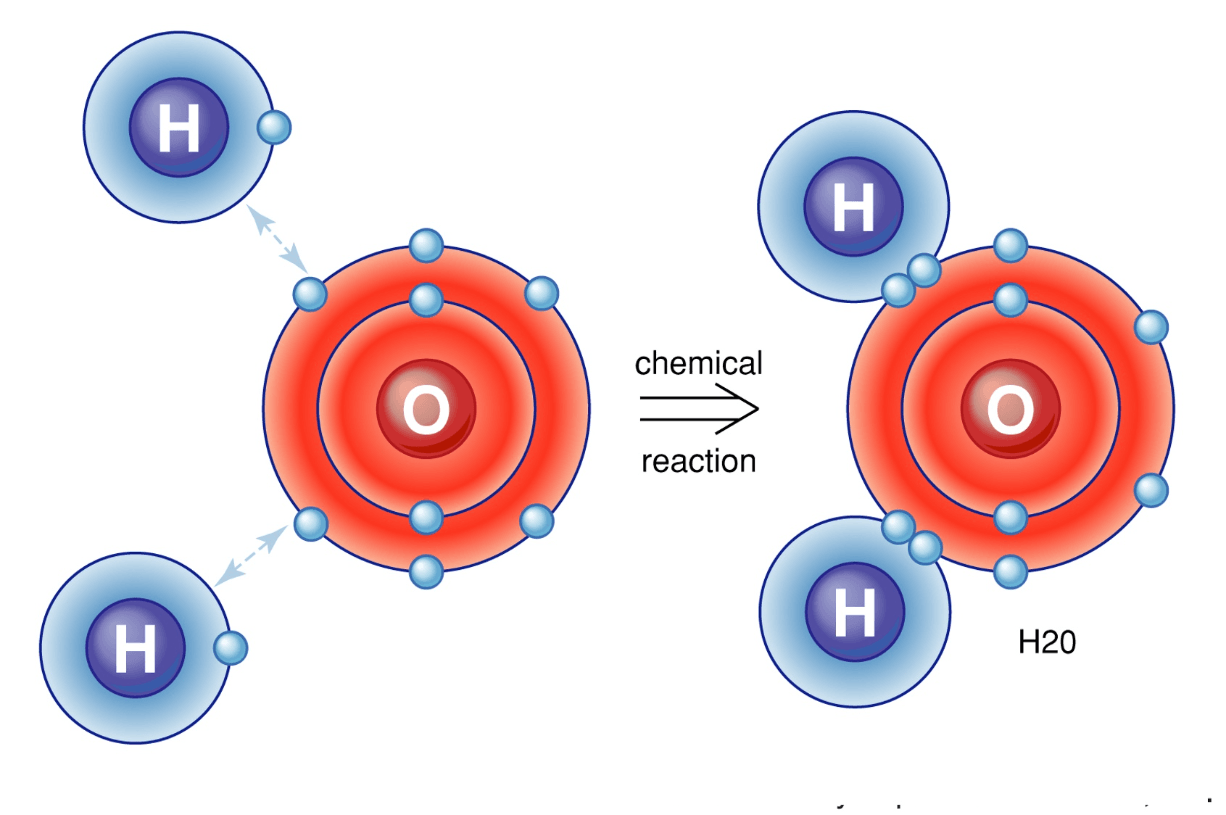
Is water (H₂O) polar or non-polar?
polar
What is the chemical formula for dichlorine heptasulfide?
Cl₂S₇