The way a mixture is combined or formed
What is physically?
Elements and compounds are known as _____substances.
What is pure?
The letters in a chemical formula
What are the element symbols?

element, compound, or mixture
What is a mixture?
Compounds are ________ (chemically/physically) combined.
What is chemically?
The two types of mixtures
What is heterogeneous and homogeneous?
True/False Compounds are different elements that are chemically combined.
What is true?
The number to the bottom right of an element symbol
What is a subscript?

Letter B: element, compound, mixture
What is an element?
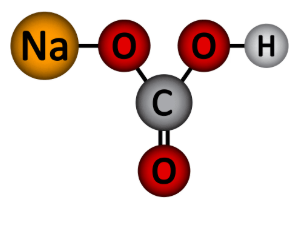
What type of molecule model is shown above?
What is the ball and stick model?
The mixture does NOT look the same all the way through (not evenly distributed)
What is heterogeneous?
The term for 2 or more atoms chemically combined, can be the same element or different elements
What is molecule?
The part of a chemical formula that tells you how many molecules you have of that compound
What is the coefficient?
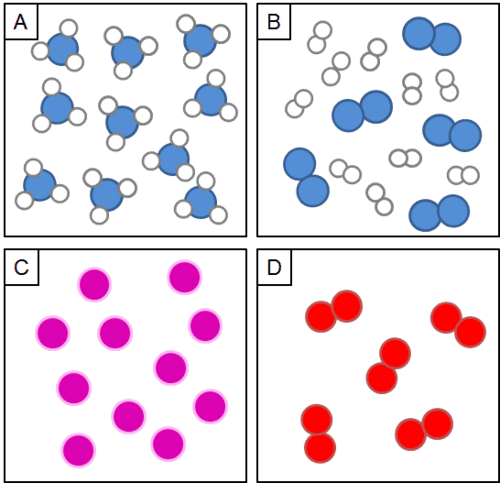
Which box letter is showing a single compound?
What is Letter A?
This represents a molecule of a compound using symbols and numbers.
What is the chemical formula?
Coffee and milk would be known as ______mixtures.
What are homogeneous?
True/False Elements that combine together to form a compound lose their original properties to become a new substance.
What is true?
The number of oxygen atoms in the compound below:
4H2SO4
What is 16?

Which box is showing a mixture?
What is box D?
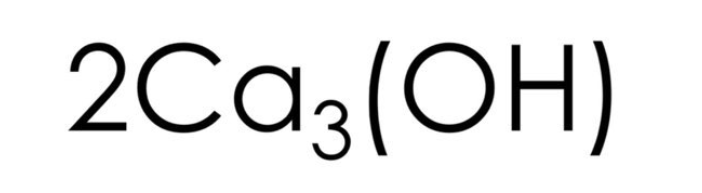
What number is the coefficient in this formula?
What is 2?
air, salad, pizza, salt water, dirt, coffee
What is air, salt water, and coffee?
From this list, these are the compounds:
CO2 N2 Air CaCl3 O3 Dirt
What is CO2 and CaCl3?
The number of phosphorus atoms in the compound below:
4Mg2(PO4)2
What is 8?
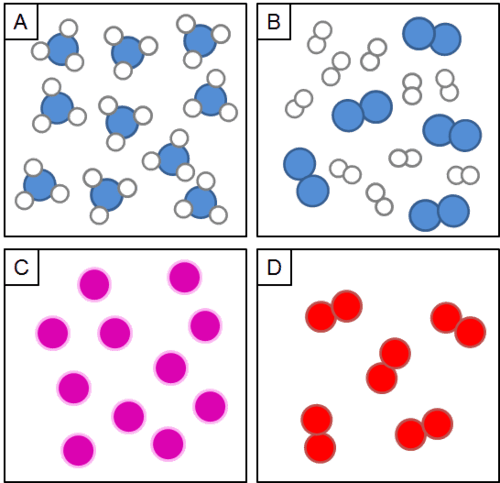
Identify the type of substance in EACH box above:
What is Box A: compound, Box B: mixture, Box C: element, Box D: element?

The TOTAL number of atoms in the formula above:
What is 8?