In an open system, what can enter or leave the system?
matter
The smallest part of a substance that cannot be broken down chemically
Atom
The 3 states of matter
solid, liquid, gas
In a closed system the mass of a chemical reaction will decrease
false
What is a mixture?
A combination of two or more substances that are physically combined and can be separated.
What does the Law of Conservation of Mass say?
Mass cannot be created or destroyed, only changed in form.
The 3 subatomic particle that make up an atom
protons, neutrons, electrons
Draw a Model of a gas

When a chemical reaction occurs new matter is created? True or False, explain if false
False, the matter is transformed into a new substance
What type of mixture looks the same throughout — like salt water?
A homogeneous mixture or solution.
During a chemical reaction, the total mass of the reactants equals the total mass of the
products
This subatomic particle has a negative charge
electrons
matter is made up of tiny ____________
particles
a substance that is present at the beginning of a chemical reaction and undergoes a change to form a new substance
reactant
In a solution, what do we call the substance that gets dissolved?
The solute.
Give an example of an open system you see every day.
Answers may include: a boiling pot of water, a living organism, or the Earth’s atmosphere.
Where are protons and neutrons found in an atom?
nucleus
What happens to the particles of a substance when it changes from a solid to a liquid?
the spread apart and move faster
characteristics of a substance that don't change. Give one example
Properties
What is the difference between a heterogeneous and a homogeneous mixture?
A heterogeneous mixture has visibly different parts, while a homogeneous mixture looks the same throughout.
In a closed system, if you mix vinegar and baking soda and trap the gas, how will the mass change after the reaction?
The mass will stay the same, showing conservation of mass.
Draw a model of an atom with labels or a key
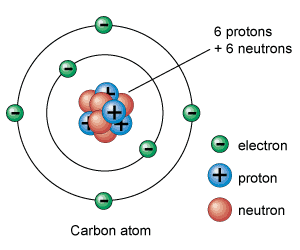
What do we call the process when a liquid turns into a gas?
evaporation or boiling
When you mix sugar in water and it dissolves, is this a chemical or physical change — and why?
It’s a physical change because the sugar’s identity doesn’t change; it can be recovered by evaporating the water.