Is it safe to taste chemicals in a lab?
No. It is not safe to taste chemicals in a lab because they may be toxic, corrosive, or unknown, and even small amounts can cause poisoning, burns, or serious health harm.
Name the three main subatomic particles.
It is the proton, neutron, and electron.
Explain what the aim of a scientific report is used for.
It states what the experiment is trying to find out.
What is an ion, and how is it different from a neutral atom?
An ion is an electrically charged particle that forms when an atom gains or loses electrons, whereas a neutral atom has an equal number of protons and electrons.
What is an isotope?
Atoms of the same element with the same number of protons but different numbers of neutrons (which makes the mass heavier or lighter).
Naturally occurring Carbon comes in three different isotopes: Carbon-12, Carbon-13, and Carbon-14.
- They all have 6 protons (that's what makes them Carbon).
- They all have 6 electrons (so they are neutral).
- However, they have 6, 7, or 8 neutrons, which gives them each a different mass
List four emergency safety items in the science classroom.
A fire extinguisher, fire blanket, eyewash station, and first aid kit.
What is the maximum number of electrons in the second shell?
8 electrons.
Remember:
First shell = maximum of 2 electrons.
Second shell = maximum of 8 electrons.
Third shell = maximum of 8 electrons.
What is the independent variable?
A student changes the amount of sunlight a plant receives and measures the height of the plant after 2 weeks.
IV = Amount of sunlight.
Reasoning:
IV is the variable that you change on purpose in an experiment. The DV is what is measured.
Describe a cation and explain how it is formed.
A cation is a positively charged ion formed when an unstable atom loses one or more electrons from its outer shell.
*Remember that t in cation is similar to a +.
- The number of positive protons stays the same.
- The number of negative electrons decreases.
- There are now more positive charges than negative charges remaining in the atom
True or False.
An isotope is electrically neutral.
True.
*An atom is also neutral. However, an ion is electrically charged.
Reasoning:
An isotope is defined by having the same number of protons but a different number of neutrons.
Charge depends on protons (+) and electrons (−), not neutrons.
In a neutral atom, the number of protons = electrons, so the charges cancel out.
Why is it a safety requirement to tie back long hair and avoid loose clothing in the laboratory?
Long hair and loose clothing must be secured to prevent them from catching fire when working near flames or heating devices.
What is the difference between an electron, a neutron and a proton?
Protons carry a positive electrical charge (+). They are located inside the tiny, dense nucleus at the centre of the atom. The number of protons is the atomic number, which defines which element the atom belongs to.
Neutrons have no electrical charge (they are neutral). Like protons, they are found in the nucleus. A neutron has almost the same mass as a proton, and the two particles together determine the atom's mass number.
Electrons carry a negative electrical charge (-). They are much smaller than protons and neutrons, possessing almost no mass. Instead of being in the nucleus, electrons move around it in fixed orbits called shells.
What are the independent variable (IV) and the dependent variable (DV)?
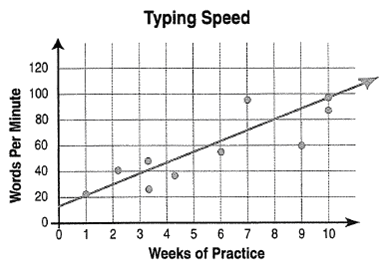
Independent Variable (IV): Weeks of practice
Dependent Variable (DV): Words per minute (typing speed)
Why do atoms gain or lose electrons to form ions?
Most atoms achieve stability by having a full outer shell of electrons; they gain or lose electrons due to collisions or other interactions to reach this stable state.
What happens if an isotope is unstable?
It becomes radioactive. Also known as radioisotopes.
Name three types of safety equipment used in Science.
Some examples:
Safety goggles (eye protection)
Lab coat / protective clothing
Gloves (e.g. nitrile gloves)
Face shield
Closed-toe shoes
Mask / respirator (for fumes or dust)
Hair tie / hair restraint (to keep hair secure)
Ear protection (earmuffs or earplugs, when needed)
Calculate how many electrons magnesium has in its 3rd shell.
There are 2 on its outermost shell.
There are 12 electrons in the shells. (2 and 8 on the first and second shell, and 2 on the last shell.)
Remember:
First shell = maximum of 2 electrons.
Second shell = maximum of 8 electrons.
Third shell = maximum of 8 electrons.
A graph shows that as the independent variable increases, the dependent variable also increases in a consistent upward straight line.
What does this relationship suggest about the variables?
This suggests a positive relationship, meaning the dependent variable increases as the independent variable increases.

How does an atom, such as Chlorine (Cl), become a stable anion?
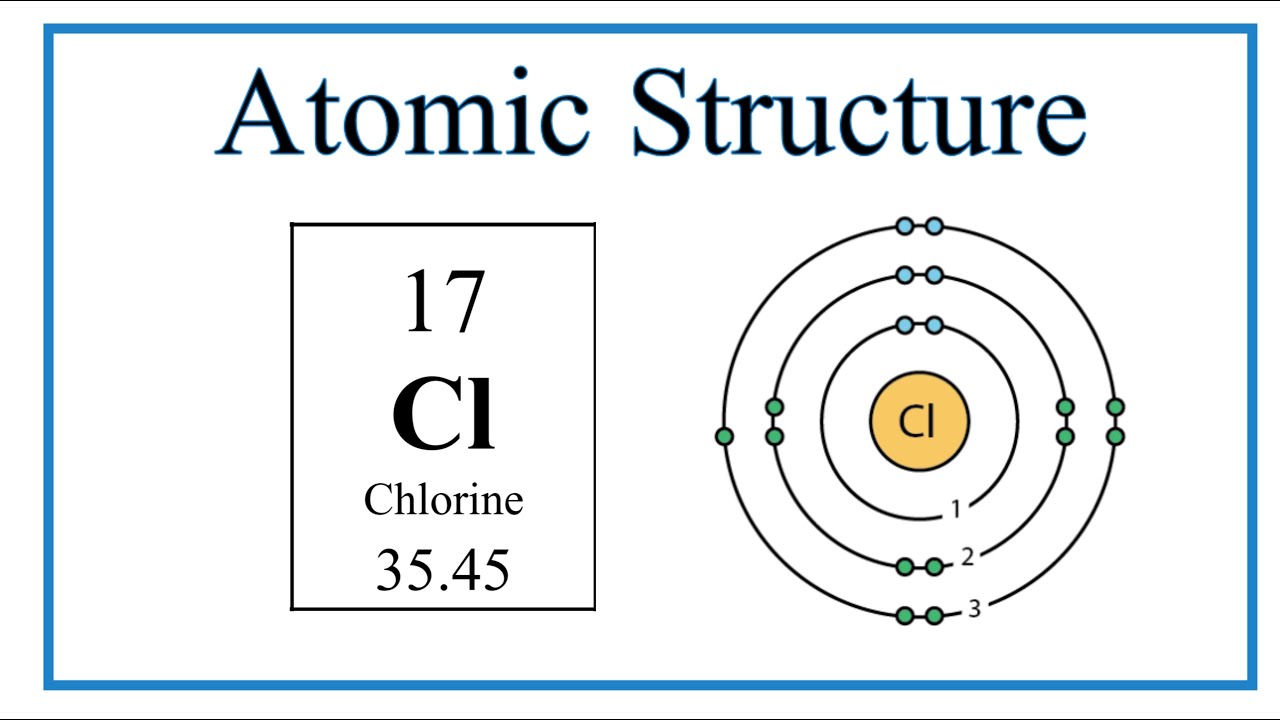
It gains one extra electron to fill its outer shell, which gives it a negative charge of -1.
In the example Chlorine-35, what does the number '35' tell us about the atom's characteristics?
It is the mass number, which is the total count of protons and neutrons added together, which in this case is 35.
Adding 17 protons and 18 neutrons together gives you a total mass of 35 in the element Chlorine.
While every atom of chlorine must have 17 protons (atomic number) to keep its identity, its mass number can change if the number of neutrons in the nucleus changes. For example, Chlorine-35 has 18 neutrons, whereas Chlorine-37 has 20 neutrons. Because these atoms are the same element but have different masses, they are called isotopes
What is the very first thing you should do if you break glassware or spill a chemical?
You must notify your teacher immediately so they can handle the cleanup or provide first aid.
If an atom has 11 protons and 12 neutrons, what is its mass number?
P + N = M
23
P + N = M
11 + 12 = 23
Adding the protons and the neutrons creates the mass number. This equation is for the element Sodium (Na).
A student is investigating how changing the temperature of a liquid affects the speed of its particles. They know that heating a substance provides it with kinetic energy. Write a testable hypothesis for this investigation using the mandatory 'If… then… because…' sentence structure.
Hypothesis: If the temperature of a substance increases, then the speed and frequency of particle collisions will increase, because the particles gain kinetic energy.
- The "If" (Independent Variable): This is the factor you are changing, which in this case is the temperature.
- The "then" (Dependent Variable): This is the factor you are measuring or observing, which is the speed and frequency of collisions.
- The "because" (Scientific Reasoning): This provides the justification for your prediction, correctly identifying that heat adds kinetic energy to particles.
A Magnesium atom (Mg) has 12 positive protons and 12 negative electrons. To become stable, it loses 2 electrons from its outer shell. What is the resulting electrical charge of the magnesium ion, and is it a cation or an anion?
It becomes a positive ion (cation).
The resulting charge is +2 (or 2+) and it is a cation
Reasoning:
The atom has fewer negative charges (electrons) than positive charges (protons).
This leaves an overall positive charge.
True or False:
Isotopes are atoms with the same number of neutrons, but different numbers of protons.
False:
Isotopes are atoms with the same number of protons, but different numbers of neutrons.
Reasoning:
The number of protons determines the element (e.g., carbon always has 6 protons).
If the number of protons changes, it becomes a different element, not an isotope.
Isotopes keep the same element identity but vary in neutrons, which changes their mass.