Brain Puzzles 1
Brain Puzzles 2
Brain Puzzles 3
Brain Puzzles 4
Brain Puzzles 5
Brain Puzzles 6
100

Travel Overseas
100
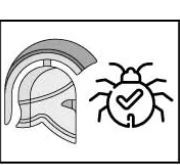
romantic
100

I believe in you
100
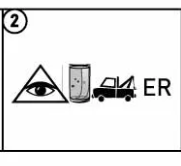
Eiffel Tower
100
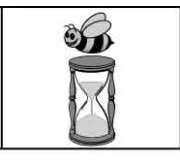
Be on time
100

Forbidden Fruit
100

Breakfast
100

A chain of events
100
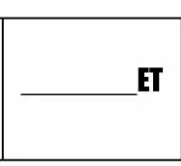
Blanket
100

Left for dead
100

Way to go
100
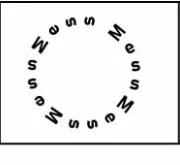
Mess around
100

Downtown
100

Forget it
100
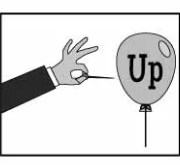
Pop Up
100

Sixth Sense
100
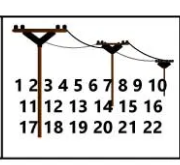
Phone Numbers
100
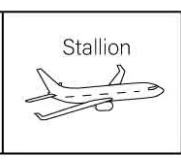
HorseFly
100
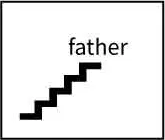
Stepfather
100
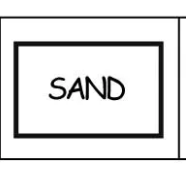
Sandbox
100

Top Secret
100
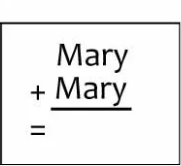
Summary
100
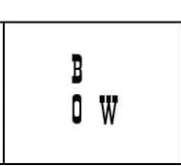
Elbow
100
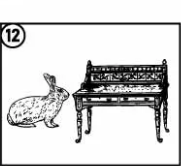
Hairdresser
100

Robin Hood
100

On second thought
100

Holy Man
100
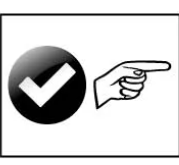
Check Point
100

Love at first sight
100
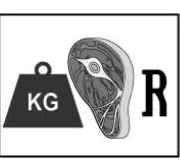
Kilometer
100
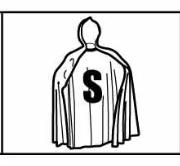
Escape
100
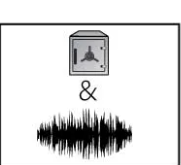
Safe and Sound
100
July
100

Double Agent
100
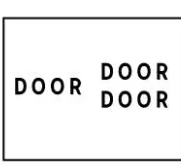
Door to door
100
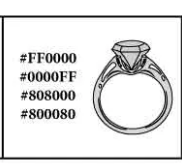
Coloring