_____________ cannot be physically separated
Pure Substance
The number of protons an atom has is it's _______
Atomic Number
4/2He Represents
An alpha particle
In a(n) ______________ bond, electrons are shared
Covalent
Alkanes must contain all _______________ bonds between carbons.
Single
The atomic model proposed by John Dalton in which atoms are solid spheres that cannot be divided into small particles is ___________________
The solid sphere model
_____________________ cannot be physically or chemically separated
Elements
The exact number of protons plus neutrons in an atom is it's ________________
Mass Number
A beta particle is the equivalent of a(n) _________
Electron
In a(n) ________________ bond, electrons are transferred
Ionic
Alkenes must contain at least one ____________ bond between carbon atoms.
Double
The atomic model proposed by JJ Thomson in which small, negatively charged electrons float in a positively charged fluid is the
Plum Pudding Model
A _____________ mixture looks uniform throughout
homogeneous
Isotopes are ________________________
Atoms of the same elements with different mass numbers
Half-life is the amount of time a radioactive atom takes to ___________________
Reduce by half
In a(n) _____________ bond, electrons move freely
Metallic
The following structural formula is an example of an alkane, alkyne, or alkene

Alkyne
The first atomic model that featured a positively charged nucleus surrounded with negatively charged electrons is called
The nuclear model
Sand is an example of a ____________ mixture
heterogenous
What charge would an ion of Oxygen have?
2-
The half-life of carbon-14 is
5715 years
In a double covalent bond, how many electrons are shared?
4
What is the functional group in the following molecule?
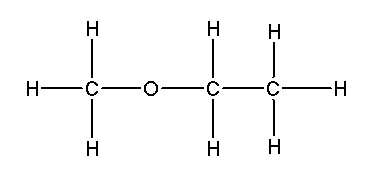
An Ether
The atomic model in which electrons orbit the nucleus at different distances is called the
Solar System Model
Homogeneous mixtures can be separated through
Evaporation/boiling
Sulfate is a polyatomic ion. What is its chemical formula?
SO4
What type of particle does Neon-19 emit?
A positron
What kind of bond would form between Lithium and Sulfur
Ionic
What is the functional group in the following molecule?
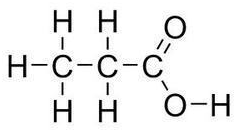
Organic Acid
The first atomic model to feature proton instead of one solid positive nucleus is the
Proton Model
In order to separate a mixture of pure water and sand, you would use _____________
Filtration
In terms of subatomic particles, explain howCarbon-14 and Carbon-12 are different.
Carbon-14 has two more neutrons than Carbon-12
You have a 100 g sample of potassium-42. After 24.72 hours, how much would be left?
25 g
What two types of bonds are involved in sodium sulfate, Na2SO4?
Ionic and Covalent
What two functional groups are present in the following molecule?
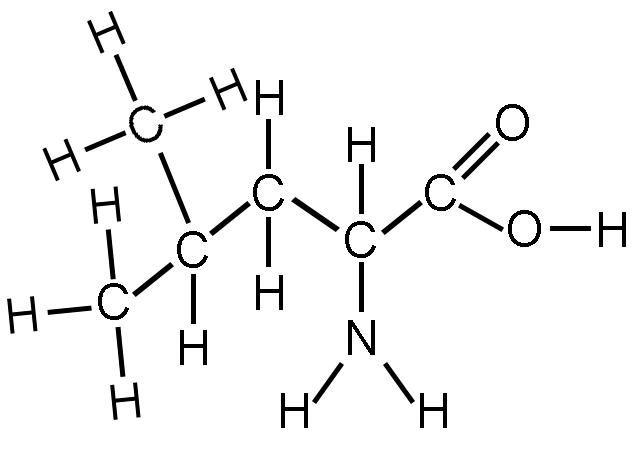
Organic acid and amide
This atomic model has electrons located in areas of probability around the nucleus called orbitals
The Wave Mechanical Model