The amount of product is determined by what type of reactant
Limiting reactant
Why do we need to balance chemical equations?
To show they follow the law of conservation of mass
Allows us to convert between moles and grams
molar mass
The units for molarity
M or mol/L
What is the equation for percent yield?
The following reaction is what type of chemical reaction?
Pb(NO3)2+2KI→2KNO3+PbI2
Double replacement
Balance the following equation:
Mg + AlPO4 → Al+ Mg3(PO4)2
3,2,2,1
Allows us to convert between moles of one substances and moles of another
Mole ratio
Calculate the molarity if 60 moles are in a 10 L solution
6 M
Convert 5761 mL to L
5.761 L
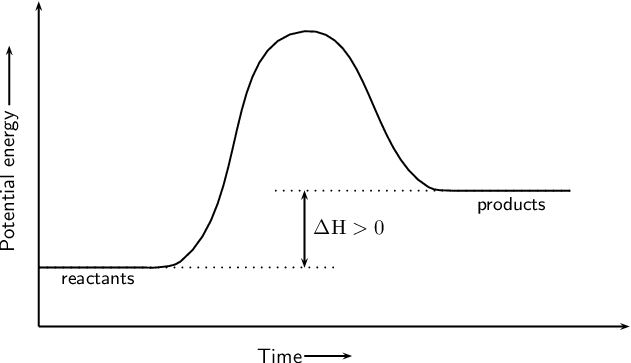 What type of reaction is shown on the graph
What type of reaction is shown on the graph
Endothermic
Balance the following equation:
CH4 + O2 → CO2 + H2O
1,2,1,2
Allows us to convert between number of particles and moles
Avogadro's number
Calculate the molarity if 4 moles are in a 10,000 mL solution.
0.4 M
Where does the mole ratio come from
(Bonus points if you write an example)
Coefficient of a balanced chemical reaction
What type of reaction is shown?
H2SO3 → H2O + SO2
Decomposition
Balance the following:
N2+O2⟶ N2O5
2,5,2
22.4 L
A solution is prepared by dissolving 42.23 g of NH4Cl into enough water to make 500.0 mL of solution. Calculate its molarity.
1.58 M
A chemist observes that a reaction beaker feels warm. What type of chemical reaction is this?
Exothermic
Predict the products of the single replacement reaction:
Mg + Al2O3→
Al + MgO
C2H6+O2⟶H2O+CO2
2,7,6,4
Allows us to convert between L of solution and moles
molarity
A chemist needs to prepare 3.00 L of a 0.250 M solution of potassium permanganate (KMnO4). What mass of KMnO4 does she need to make the solution?
119 g
What is a precipitate
A solid formed in a chemical reaction