What is the weakest intermolecular force?
Dipole-Dipole, London Dispersion, or Hydrogen bonding
London dispersion
What is one piece of evidence that a chemical reaction has occurred?
Bubbles, heat produced, light produced, precipitate (solid formed), odor, color change...
The mass of one mole of a substance is equal to the _________ ________ of that substance (from the periodic table).
molar mass
Which state of matter has a definite volume and a definite shape?
Solid
Convert 56 degrees Celsius to Kelvin.
329 K
What is the molecular geometry of CO2?
Linear
6
What is the molar mass of Cu(NO3)2?
187.56 g/mol
Gas
Convert 80 psi to atmospheres (atm)
5.44 atm
What is the molecular geometry of water a molecule?
Bent
Balance the following equation:
__Fe + __O2 --> __Fe3O4
3Fe + 2O2 --> 1Fe3O4
A student produces 25g of a compound, though the reaction should theoretically produce 81g. What is the percent yield?
30.86%
What is the boiling point of this substance based on this heating curve?
134
A gas occupies 12.3 L at a pressure of 40.0 mmHg. What is the volume when the pressure is increased to 60.0 mmHg?
8.2 L
What is a polar molecule?
A molecule with unequal sharing of electrons. These molecules experience dipole-dipole forces.
Balance AND classify the following reaction:
__Al2O3 --> __Al + __O2
2Al2O3 --> 4Al + 3O2
Decomposition
2H2 + O2 --> 2H2O
How many moles of H2O can be produced from 6 moles of O2?
12 moles of water
Which region of this phase diagram represents a gas?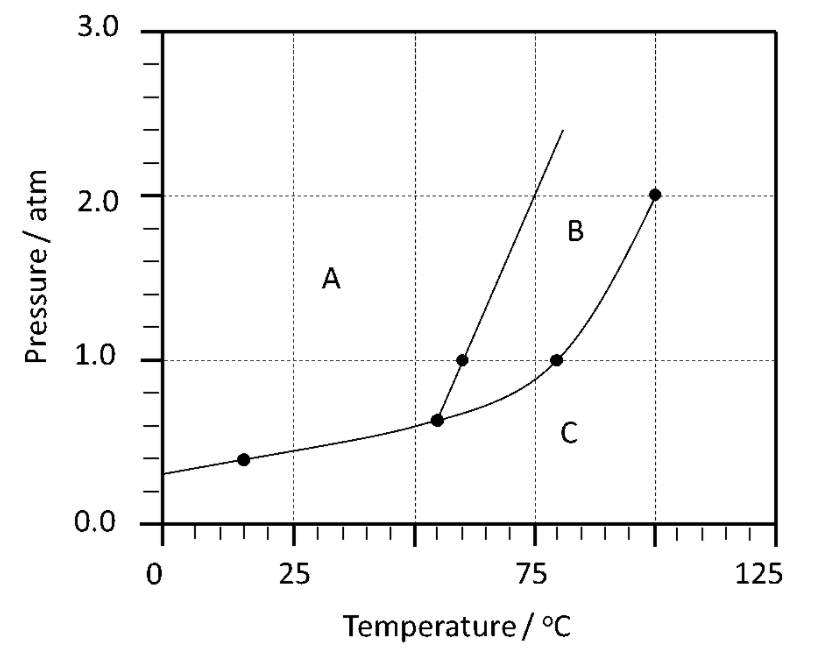
C
A balloon is filled with 2.50 L of gas at a room temperature of 22.0 C. If the balloon is placed in a warmer room where the temperature is 71.0 C, what is the new volume of the balloon?
2.92 L
The boiling point of water is 100 degrees Celsius, while the boiling point of acetone is 56 degrees Celsius. Which has weaker intermolecular forces?
Acetone
Write the following balanced chemical equation:
Lithium reacts with oxygen to produce lithium oxide.
4Li + O2 --> 2Li2O
2H2 + O2 --> 2H2O
How many grams of H2 are needed to produce 15 grams of water?
1.68 grams of hydrogen
What is the name of the phase change going from a gas to a solid?
Deposition
A 10.0 L tank contains 0.10 moles of gas at 500 K. What is the pressure in atm?
0.41 atm