Is rust an example of a chemical or physical change?
Why, name one reason.
Chemical because it is exothermic, there is a color change and new substances are formed.
What are examples of a physical changes involving water?
Melting, freezing, vaporization, condensation
What are reactants in a chemical change?
The substances that go into a chemical reaction
Heat is absorbed from the environment during this type of chemical reaction.
Endothermic
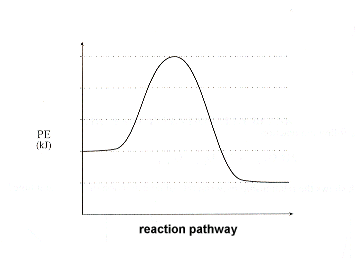
This graph indicates which type of reaction? Why?
Exothermic because the temperature rose during the reaction.
Is boiling an egg a chemical change or physical change? Name one reason why.
Is the boiling water a chemical or physical change? Why?
The egg becoming hard-boiled is a chemical change because it is the formation of a solid.
Boiling water is a physical change because it is a change in the state of matter.
What are three pieces of evidence a physical change has taken place.
1. Change in State of Matter
2. Change in size or shape
3. Substances stay the same
What are the new substances that are formed by a chemical reaction called?
Products
Heat is released during the reaction during this type of chemical reaction.
Exothermic
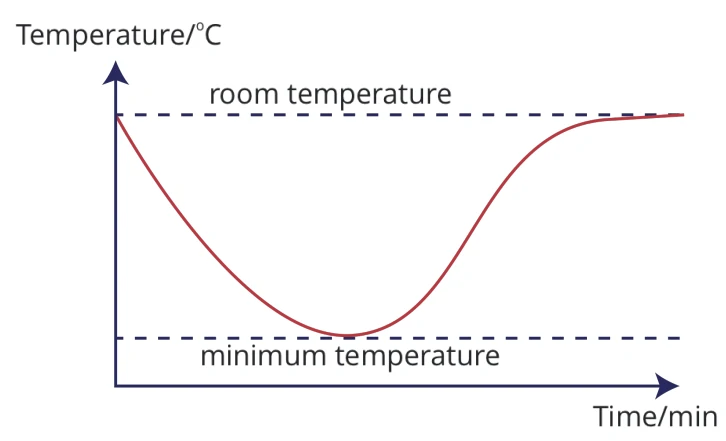
Does this graph of the temperature changes during a chemical reaction indicate that it is an exothermic or endothermic reaction?
Endothermic.
Melting chocolate to mold into a bunny is an example of a ________ __________ because it is a ________________________________________ .
What is a physical change because it is a change in the state of matter.
What is a physical change?
The material involved in the change is structurally the same before and after the change. (you can go back to your original form)
What is a chemical change?
a change of materials into another, new materials with different properties and new substances are formed
(Something can go back to its original form or can be changed back)
This type of reaction forms bonds. It is bond making.
Exothermic Reactions

Explain how this graph shows an endothermic reaction is occurring?
The temperature first drops since energy is being absorbed. It then begins to rise once the reaction is complete because heat energy transfers until it reaches thermal equilibrium (room temperature).
Is a banana rotting an example of a chemical or physical change? Name two reasons why.
Chemical change because there is a color change, an odor is produced, new substances are formed.
Is burning wood considered a Physical change?
No, because it cannot be changed back to its original form.
Heat and light are produced.
Name five pieces of evidence a chemical change has taken place.
Color change, heat and light produced, production of a gas, formation of a solid, new substances (products) are formed, cannot easily be reversed, temperature change
This type of reaction forms bonds during the reaction. This is a bond breaking reaction.
Endothermic Reaction

Why did the temperature during this reaction begin to fall and then flatten out?
The reaction was over. The reactants were all used up so the temperature dropped until it reached room temperature because heat transfers until it reaches thermal equilibrium.
Why is baking a cake an example of a chemical change? Name at least two reasons.
1. Once the cake is formed, we cannot get back the original batter.
2. Production of a solid
3. Color change
4. Production of a gas
Frost forming on the grass is an example of a chemical or physical change? Why?
Physical change.
Change of state of matter from a liquid to a solid.
How does a chemical change, change a substance?
(*hint it's something to do with the atoms and molecules)
When bonds are broken and/or formed between molecules or atoms creating new substances (products) that are different from the reactants.
During the liver lab, which of the experiments was endothermic? How do you know?
Baking soda and vinegar because the temperature dropped.

Is this an example of an endothermic or exothermic reaction? How do you know?
Exothermic because heat is being released as part of the products.
Bubbles formed during the potato and hydrogen peroxide experiment. Is this evidence of a chemical or physical change? What does this tell us about the products in this reaction?
Chemical change.
A gas is being produced.
Describe why slicing pizza is a physical change but baking a pizza is not.
When you slice pizza... you still have pizza. (The substance doesn't change.)
When you bake pizza the individual ingredients that make up the pizza change into something else.
During the splint test after the liver and hydrogen peroxide reaction, the splint began to glow and relit. What can we conclude about the products for this reaction?
Oxygen was produced as fire needs it to fuel combustion.
During the liver lab, the first splint relit when it was placed inside the test tube. Why was the second flame extinguished during the flame test?
All of the oxygen created by the reaction was used up during the splint test. The reaction was over so no more oxygen was being produced.
Why is the top reaction endothermic and the bottom reaction exothermic?
1.Heat is a part of the reactants. It is being absorbed from the environment to give the reaction the energy it needs to break bonds.
2. Heat is part of the products. It is being released to the environment as bonds form.