Matter is anything that has ____________ and takes up ____________.
Matter is anything that has mass and takes up space.
What is a mixture?
A physical blend of two or more components.
Which subatomic particles make up an atom & what their charges?
proton (+)
neutron (0)
electron (-)
What is the atomic number?
The number of protons
What are isotopes?
True or False.
Breaking a glass cup is an example of a physical change.
True
The composition of the cup did not change. It is still glass after it broke.
Label images A-D as either element or compound.

A: compound
B: element
C: compound
D: element
Which subatomic particles are located in the nucleus?
protons and neutrons
What is the atomic number of No?
102
True or false.
Isotopes of the same element have the same number of electrons.
False.
Isotopes of the same element have the same number of protons.
A chemical change will change the composition (what it is made of) of matter but a physical change will not.
What is the difference between a homogenous mixture and a heterogenous mixture?
Homogenous mixture: mixture that appears uniform (the same) throughout
Heterogenous mixture: mixture where you can see every substance within the mixture

What makes up the mass number of an atom?
What is the chemical symbol of mercury?
Hg
What is the difference between Fe-57 and Fe-54 isotopes?
Fe-57 has three more neutrons than Fe-54
What is the difference between extensive and intensive properties?
Extensive: depends on the amount of matter (mass, surface area, volume, etc)
Intensive: depends on the type of matter (flammability, boiling point, density)
Air is an example of which of the following
a. compound
b. element
c. homogenous mixture
d. heterogenous mixture
Which number represents the atomic number? 
79
How many neutrons does the isotope carbon-14 have?
8 neutrons
# neutrons = mass number - atomic number
= 14 (from hyphen notation) - 6 (from periodic table)
= 8
Are isotopes always stable?
No only a few are stable.
They will undergo radioactive decay in order to become stable .
What is the law of conservation of mass?
The mass of the reactants = the mass of the products
Image (3) consists of ______________.
Image (3): Mixture of elements
How would write this in hyphen notation?

Au-196
or
Gold-196
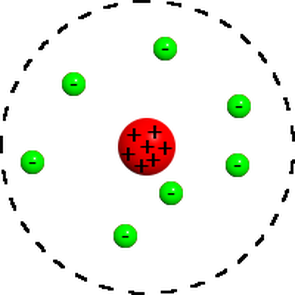
What is the difference between the plum pudding model and the nuclear model of an atom?
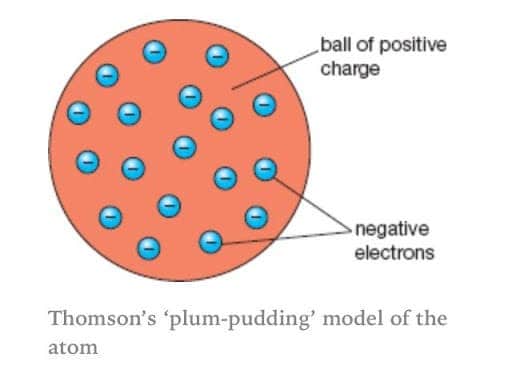
rutherford nuclear model has a positive center (nucleus)
mass number: the number of protons and neutrons in the nucleus for an individual atom/isotope
atomic number: is the average mass numbers of all known isotopes of an element
