12. Sucrose is another name for table sugar. Sucrose is a compound made from the elements carbon, hydrogen, and oxygen. Which statement best describes the properties of sucrose?
A. They are similar to properties of carbon
B. They are similar to properties of oxygen
C. They are similar to the properties of hydrogen
D. They are different from the properties of all the elements sucrose contains
D
10. Robert is hiking through the forest and writing down his observations. He steps on a branch and it breaks into several pieces. Which of the following best describes the resulting properties of the branch?
A. The density of the branch decreased, since it now takes up less space
B. The molecular arrangement of the branch changed as the broken pieces changed its state of matter
C. The total mass of the branch pieces is the same, and only a physical change has occurred
D. The chemical composition of the branch changed as the broken pieces reacted with the environment
C
14. Which of these substances is a compound?
A. Chlorine
B. Ammonia
C. Uranium
D. Nitrogen
B
8. Suppose a candle is burned in a closed system where matter cannot enter or leave. Given this situation, what is equal to the mass of the original candle?
A. The mass of the burned candle
B. The mass of all gases in the closed system
C. The mass of the gases released while the candle is burned
D. The mass of the burned candle plus the mass of the gases released while the candle is burned
D
13. How can you separate the compound that makes up sand into its elements?
A. By using a filter
B. By using a magnet
C. By melting the sand
D. By chemical changes
D
4. Which of these common substances is a homogeneous mixture?
A. Pure water
B. Table salt
C. Whole milk
D. Maple syrup
D
6. Which never changes during a physical change?
A. State of matter
B. Temperature
C. Total mass
D. Volume
C
5. A material being examined is the same throughout. The material contains carbon and oxygen, chemically combined. What type of material is it?
A. Element
B. Compound
C. Homogeneous mixture
D. Heterogeneous mixture
B
4. Which of these common substances is a homogeneous mixture?
A. Pure water
B. Table salt
C. Whole milk
D. Maple syrup
D
9. Which is true when a piece of paper is crumbled?
A. Volume and mass increase
B. Volume and mass decrease
C. Volume and mass do not change
D. Volume might decrease or increase, but mass does not change
D
14. Which of these substances is a compound?
A. Chlorine
B. Ammonia
C. Uranium
D. Nitrogen
B
13. How can you separate the compound that makes up sand into its elements?
A. By using a filter
B. By using a magnet
C. By melting the sand
D. By chemical changes
D
12. Sucrose is another name for table sugar. Sucrose is a compound made from the elements carbon, hydrogen, and oxygen. Which statement best describes the properties of sucrose?
A. They are similar to properties of carbon
B. They are similar to properties of oxygen
C. They are similar to the properties of hydrogen
D. They are different from the properties of all the elements sucrose contains
D
6. Which never changes during a physical change?
A. State of matter
B. Temperature
C. Total mass
D. Volume
C
11. Some compounds are classified as acids or bases. The pH scale shows how acidic or how basic these compounds are. The lower the pH, the more acidic a compound is. The higher the pH, the more basic it is. Sodium hydroxide, a compound commonly found in drain cleaners, has a pH of 13.

Which of these phrases best describes sodium hydroxide?
A. Highly basic
B. Highly acidic
C. Slightly basic
D. Slightly acidic
A

2. Which of the four mixtures that Kim served at dinner is classified as a suspension?
A. A
B. B
C. C
D. D
A
10. Robert is hiking through the forest and writing down his observations. He steps on a branch and it breaks into several pieces. Which of the following best describes the resulting properties of the branch?
A. The density of the branch decreased, since it now takes up less space
B. The molecular arrangement of the branch changed as the broken pieces changed its state of matter
C. The total mass of the branch pieces is the same, and only a physical change has occurred
D. The chemical composition of the branch changed as the broken pieces reacted with the environment
C

7. Which is demonstrated by the steps shown in the flow chart?
A. Addition of thermal energy
B. Change in state of matter
C. Conservation of mass
D. Creation of a compound
C
8. Suppose a candle is burned in a closed system where matter cannot enter or leave. Given this situation, what is equal to the mass of the original candle?
A. The mass of the burned candle
B. The mass of all gases in the closed system
C. The mass of the gases released while the candle is burned
D. The mass of the burned candle plus the mass of the gases released while the candle is burned
D
9. Which is true when a piece of paper is crumbled?
A. Volume and mass increase
B. Volume and mass decrease
C. Volume and mass do not change
D. Volume might decrease or increase, but mass does not change
D
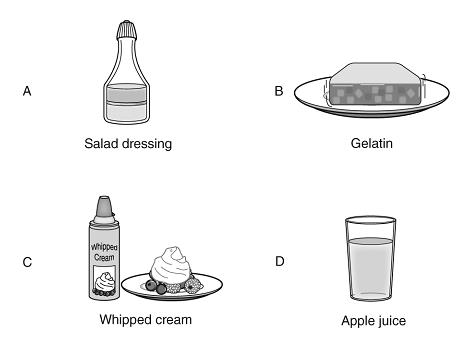
1. The four items shown above were served at a dinner at Kim’s house. Each of the items is a mixture. Kim turned on a flashlight and pointed it at each of the four items. Which of the items did the light pass through without changing?
A. A
B. B
C. C
D. D
D
3. A magnet was placed near a pile that contained both iron and sulfur. The magnet was moved gradually closer to the pile. As it neared the pile, the magnet started attracting small pieces of iron from the pile.

Which of these statements best describes the contents of the pile?
A. It is a homogeneous mixture of iron and sulfur
B. It is a heterogeneous mixture of iron and sulfur
C. It is a compound that contains both iron and sulfur
D. It is a compound that can be separated by magnetism
B
4. Which of these common substances is a homogeneous mixture?
A. Pure water
B. Table salt
C. Whole milk
D. Maple syrup
D
5. A material being examined is the same throughout. The material contains carbon and oxygen, chemically combined. What type of material is it?
A. Element
B. Compound
C. Homogeneous mixture
D. Heterogeneous mixture
B
15. The pH scale shows whether a compound is acidic, basic, or neutral. Sodium chloride, which is table salt, is neutral. Neutral compounds are neither acidic nor basic.


From the scale, what is the pH of sodium chloride?
A. 0
B. 7
C. 10
D. 14
B