How can you tell the difference between ionic and covalent compounds?
two nonmetals = covalent
metal and nonmetal = ionic
What is Electronegativity?
The ability of an atom to attract electrons toward itself
Compound with this type of bond are typically found as solids at room temperature.
Ionic Bonds
The formula for chlorine dioxide
ClO2
What is an isomer
Compounds with the same molecular formula but different structure (arrangements of the atoms)
Octet Rule
What is the rule that states atoms form compounds to achieve 8 valence electrons?
What are the two types of covalent bonds?
Polar Covalent Bonds Non-polar Covalent Bonds
Compound with this type of bond have very low melting points (sometime below room temperature).
Covalent Bonds
The name of O2
Oxygen gas or molecular oxygen
T/F All isomers of the same element will have the same types of covalent bonds
False
How do atoms obey the octet rule differently when forming ionic vs covalent compounds?
Ionic = steal electrons
Covalent = share electrons
If permanent marker gets on your clothes, what must you use to get it out?
Compound with this type of bond are good at conducting electricity when dissolved.
Ionic bonds
The name of CH4
Carbon Tetrahydride
bonus point for methane
Identify how many of each element is in the diagram.
H = 12
C = 6
Which type of bond is the strongest?
triple bond
What characteristic makes an ionic bond instead of a covalent bond?
A big difference in the electronegativity of the atoms.
Which type of compound is more likely to be soluble in ethanol?
The name of Si2Br6
Disilicon Hexabromide
How many hydrogen are in the image.
24
What determines the bond length of a covalent bond?
Potential energy based on the attraction of the nuclei to the shared electrons
When is a a covalent bond formed instead not an ionic bond?
When the difference in electronegativity is below 1.7.
Compound with this type of bond are good at conducting heat and electricity in a solid form.
Metallic bonds
Br6Si
FINAL JEOPARDY
Draw an isomer of the following compound.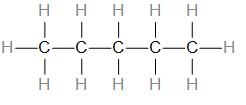
:)