All elements in the periodic are listed in order of increasing this.
What is atomic number?
(Or, what is number of protons?)
Two atoms which have the same number of ________ but different numbers of _________ are called isotopes.
What are:
- same number of protons
- different number of neutrons
This is the name of the type of electromagnetic radiation which has the most amount of energy.
What are gamma rays?
The electrons located in the outermost shell of an atom are called this.
What are valence electrons?
This is to what the phenomenon of "ionization energy" refers.
What is the energy necessary to remove one electron from an atom (in the gas phase)?
Of the elements listed below, atoms of this one have the smallest atomic radius:
- Helium
- Oxygen
- Sodium
- Boron
What is Helium?
The symbol of this element is W.
What is tungsten?
This is the name given to the elements listed in the last column on the right of the periodic table.
What are the noble gases?
This is the number of electrons in an atom of uranium.
What is 92?
A wave of a red light has a (higher/lower) frequency than a wave of violet light.
What is a lower frequency?
This is the element with the following electron configuration:
1s22s22p63s23p64s13d5
What is Cr (chromium)?
This is one possible element in the same period as sulfur which has a higher ionization energy than sulfur.
What is (either) chlorine (or) argon?
When comparing atomic radius and ionic radius of a given element, a positive ion should be expected to be [bigger / smaller] than an atom of that element.
What is smaller?
This is another name for a vertical column of elements on the periodic table.
What is a group?
The orbital "block" which contains the elements boron through neon is called this block.
What is the "p" block?
This is the number of electrons in a Na+1 ion.
What is 10?
This is the speed at which all electromagnetic radiation travels.
What is the speed of light?
(Or, approximately 3 x 108 m/s)
If the last (and highest-energy) electron of an atom is the second electron placed in a 6p orbital, then the atom must be this element.
What is lead (Pb)?
This is the reason that Cs has a lower ionization energy than K.
What is: "the outermost electron in Cs is farther away from the nucleus than the outermost electron in K, which makes the Cs electron easier to remove"?
This is the technical reason that atomic size decreases when looking at elements from left to right in the same period.
What is increasing effective nuclear charge?
This is the lightest subatomic particle.
What is an electron?
The periodic table contains elements which are all considered metals, nonmetals, or this.
What are metalloids?
If element X has three isotopes - 816X, 818X, and 820 X - which have identical percent abundances, then this is the average atomic mass of element X.
What is 818 amu?
This is the term used for electromagnetic radiation which has enough energy to blast electrons away from atoms or molecules.
What is ionizing radiation?
The abbreviated electron configuration for tin (Sn) is this.
[Write on board]
What is:
[Kr] 5s2 4d10 5p2
This is the reason all Group 1A elements have low ionization energies.
What is: "it is energetically favorable for these elements to lose one electron"?
(Or "shielding causes particularly low effective nuclear charge in Group 1A elements, but effective nuclear charge increases going left to right in a period")
(Or something like: "they want to lose one electron anyway")
This is the technical phrase which is the reason atoms of elements going down a group on the periodic table are progressively larger.
What is "shielding"?
On a scale of 0.0 to 4.0, the electronegativity of the noble gases is this.
What is 0.0?
The name given to the elements in the "d-block" area of the periodic table is this.
What are the transition metals?
If the two isotopes of chlorine (35Cl and 37Cl) yield an average atomic mass of 35.45 amu, then the percent abundance of each isotope is this.
[Write answers on board]
What is:
35Cl = 77.5%
37Cl = 22.5%
A photon of violet light (400. nm) has this energy (in Joules).
[h = 6.626x10-34 J s; c = 2.998x108 m/s)]
[1 m = 1x109 nm]
[Write answer on board; sig figs matter!]
What is 4.97x10-19 J?
The complete orbital diagram for arsenic (As) is this.
[Write on board]
What is:
This is the reason why noble gases have extremely high ionization energies.
What is "because the noble gases have full valence shells, and therefore have no benefit to gaining/losing electrons"?
(Or at least: "because the noble gases are very stabile"?)
This is the reason a phosphorus ion is so much larger than a silicon ion.
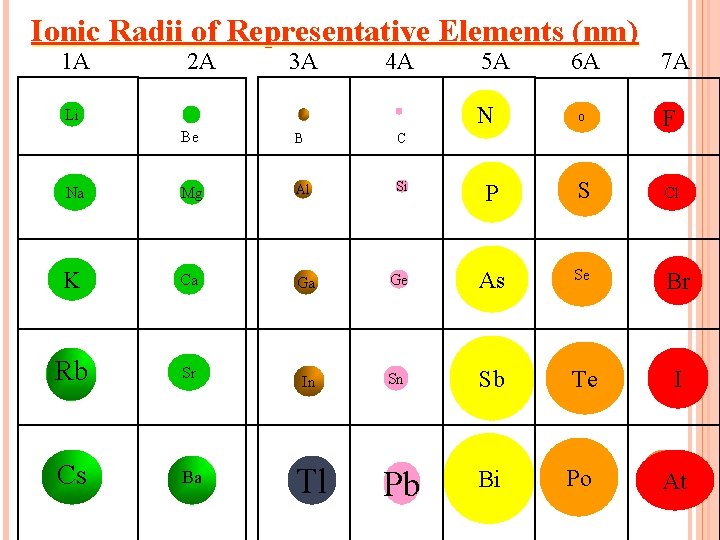
What is: "phosphorus becomes a negative ion (-3), while silicon becomes a positive ion (+4)"?
The Rutherford gold foil experiment was an improvement upon the commonly-accepted "plum pudding" atomic model because it demonstrated these two facets of atomic structure.
What is:
1) atoms were mostly empty space
2) atoms have a dense nucleus