All matter has _____ and _____.
Mass and Volume
We measure mass using a _______ and the units are ______.
Scale
Grams
The elevator on the ____ has more density because ____.
Right
It has more people in the same area (Or the people are more packed together)
The density of water is ____.
1 g/mL
Name the states of matter from coldest to hottest.
Solid
Liquid
Gas
The force that pulls more dense materials down is _____.
The amount of matter in an object
Mass
We measure volume using a ______ and the units are ______.
Graduated Cylinder
milliLiters (mL)
The _____ has a higher density because ______.
The baseball has a higher density because it has more mass in the same volume. (Or the particles are more packed together)
Objects with a density more than 1 g/mL will _____, but objects with a density less than 1 g/mL will _____.
Sink
Float
We can change the state of matter by changing the ______.
Temperature
In the water cycle, the heat comes from _____, but in the mantle cycle, the heat comes from _____.
The Sun
The Core
The amount of space an object takes up
Volume
We measure density using a _____ and the units are _____.
g/mL
These boxes have the same ______, but Box B has more _____.
Same Volume
More Mass and Density
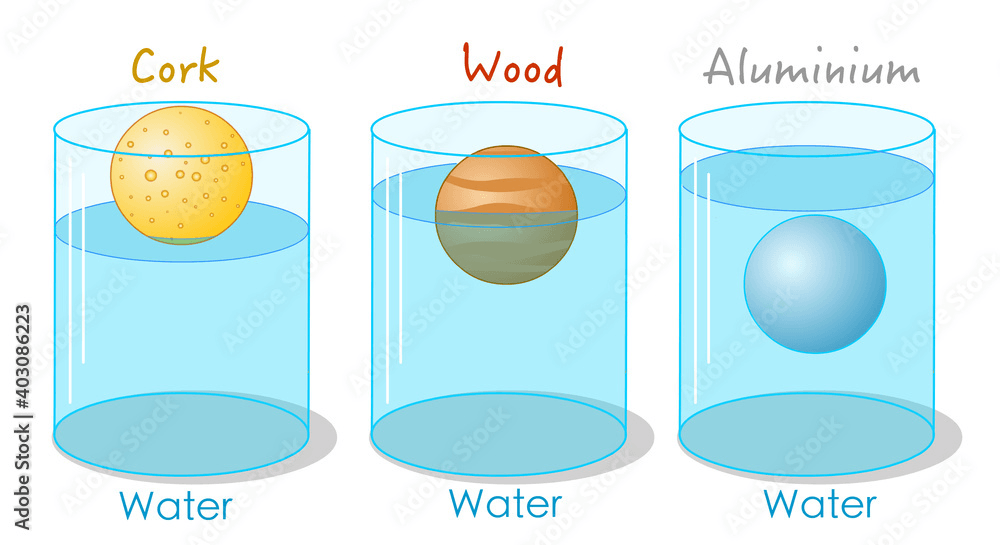
What can we infer about the density of:
- Cork
- Wood
- Aluminum
- Cork - Less than 1 g/mL
- Wood - Equal to 1 g/mL
- Aluminum - More than 1 g/mL
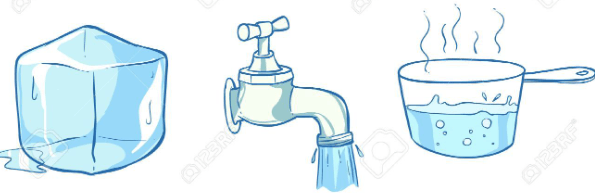
Describe the states of matter you observe.
The ice cube is a solid.
The water in the faucet is a liquid.
The water in the pot is a gas.
In both the water cycle and the mantle cycle, particles ____ and ____ when they heat up because density ____.
Spread Out and Rise
Decreases
When we talk about waves, volume means ______, but when we talk about matter, volume means _____.
How loud or quiet something is
The amount of space an object takes up
The formula for density is _____ divided by ______.
We know we wrote it right if we drew a ______.
Volume
Heart
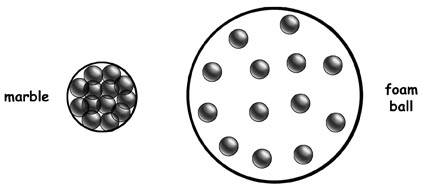 These balls have the same ______, but the marble has _______ and ______.
These balls have the same ______, but the marble has _______ and ______.
Same Mass
Marble has less volume and more density
Why do we need to stir peanut butter before we eat it?
The parts will separate by density. The peanuts will move to the bottom and the oil will rise to the top.
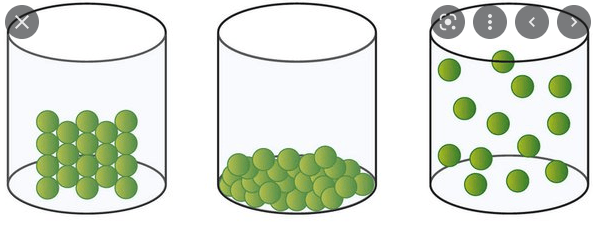
What states of matter do you observe? How do you know?
The first beaker is a solid because the particles are packed close together.
The middle beaker is a liquid because the particles are not packed close together or spread apart.
The final beaker is a gas because the particles are very spread apart.
In both the water cycle and the mantle cycle, particles ____ and ____ when they cool down because density ____.
Pack Together and Sink
Increases
Density is the amount of ______ in a specified ______.
Mass
Volume
A marble has a volume of 5 mL and a mass of 20 g.
What is the density?
4 g/mL
These boxes have the same _____, but the blue box has _____ and _____.
Same Mass
More volume and less density
You have three liquids:
- Water
- Blood (1.6 g/mL)
- Alcohol (0.8 g/mL)
You pour them into a cup and leave it overnight. What do you expected in the morning?
- The Blood will be on the bottom
- The water will be in the middle
- The alcohol will be on top
What happens to the density of materials as their temperature increases?
As temperature increases, density decreases because the particles spread out.
In the water cycle, particles cool when they touch _____, but in the mantle, particles cool when they touch ____.
The atmosphere
The crust
