The electron configuration for F
what is 1s2 2s2 2p5?
The element for [Ne] 3s2 3p4
What is sulfur?
The Lewis Model for potassium.
What is K with 1 valence electron?
What is valence electron from left to right of periodic table -group 1, group 2, group 13, group 14, group 15, group 16, group 17, group 18
1, 2, 3, 4, 5, 6, 7, 8
Number of rings on the Bohr Model for silicon.
What is 3?
The element that has a configuration of 1s2 2s2 2p6 3s2 3p6
what is Ar?
Noble gas configuration for Cobalt.
[Ar] 4s2 3d7
One way to get points off of your Lewis model drawing.
What are dots sticking straight off your drawing- (NOT HUGGING the symbol)?
Sketch what the periodic table actually looks like.
See under the newline board.
Number of protons, neutrons, electrons in the nucleus for tin.
What is protons= 50? What is neutrons= 69? What is electrons= 50?
What is Nickel
The noble gas configuration of Sn
True or false.
The Lewis dot diagram for I has 1 valence electron.
Describe how to use the periodic table to find the energy and valence electrons of an element.
What is the period equals the energy level/number of rings on the Bohr model.
What is the group number indicates the number of valence electrons/dots around the Lewis dot diagram?
Draw the Bohr model for Be.
Be
The electron configuration of Sn
What is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p2
Noble Gas configuration of Po.
What is Po- [Xe]6s24f145d106p4
As forms this many bonds.
What is 3?
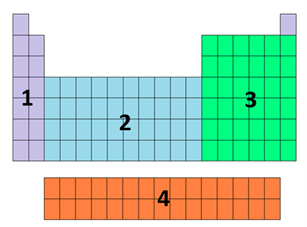
the block represented by each number
1 = s
2 = d
3 = p
4 = f
Draw the Bohr model for Cl.
Cl
Electron Configuration for Ba
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2
Noble gas configuration for Fe
[Ar] 4s2 3d6
Lewis model for bromine.
What is Br with 7 valence electrons?
The challenge about period 4, 5, 6 and 7 when doing electron configurations.
4s drops 1 energy level to 3d.
6s drops 2 energy levels to 4f, back up to 5d, and finally back to 6p.
Draw the Bohr model for Te.
Te