This is the process in which substances interact and form new substances.
What is a chemical reaction?
A reaction that releases energy to its surroundings is called this.
What is exothermic?

What is an exothermic reaction?
A candle burns and gives off heat and light. What does this show?

That a chemical reaction is releasing energy.
In a chemical equation, the substances on the left side are called these.
What are reactants?
Exothermic reactions absorb energy from their surroundings.
Fix it: Exothermic reactions release energy to their surroundings.
These are the starting substances in a chemical reaction.
What are reactants?
A reaction that stores energy or takes in energy from its surroundings is called this.
What is endothermic?

What is an endothermic reaction?
A student mixes vinegar and baking soda and sees bubbles form. The bubbles are evidence of what?
What is gas production, showing a chemical reaction?
In a chemical equation, the substances on the right side are called these.
What are products?
“If a reaction gets colder, it must be exothermic.”
Fix it: If a reaction gets colder, it is likely endothermic.
These are the new substances made during a chemical reaction.
What are products?
A hand warmer gets hot after the chemicals inside are mixed. This is what kind of reaction?
What is an exothermic reaction?
A student records bubbles, a drop in temperature, and a new odor after mixing two substances. Which piece of data best supports that a chemical reaction occurred?
What are bubbles and temperature change?
(Any answer naming valid evidence of reaction is fine.)
A hot pack becomes warmer after chemicals are mixed. Explain what happened to the energy.
The reaction released energy to the surroundings, making the pack feel warmer.

What type of reaction is photosynthesis?
Endothermic reaction. Energy is on the reactant side.
“Bubbles always mean a physical change.”
Fix it: Bubbles can be evidence of a chemical reaction because a gas may be produced.
Name two signs that a chemical reaction may have happened.
What are temperature change, color change, gas production, light produced, odor change, or precipitate formed?
An instant cold pack gets cold when chemicals inside it are mixed. This is what kind of reaction?
What is an endothermic reaction?
A graph shows temperature rising quickly after two powders are mixed. What can you conclude about the reaction?
That the reaction released energy and was likely exothermic.
Muffins baking is an example of what type of reaction? Endo or Exo?
Endothermic. The muffins are absorbing the heat from its surroundings.
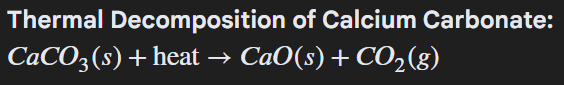 Thermal Decomposition of calcium carbonate is what type of reaction.
Thermal Decomposition of calcium carbonate is what type of reaction.
Endothermic. The heat energy is on the reactant side.
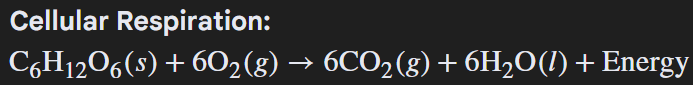
Cellular respiration is an example of an endothermic reaction
FIX IT: energy is on the product side which means it is an Exothermic reaction.
Why is a temperature change during a reaction important evidence in PS3.1?
Because it shows the reaction may have released energy or stored energy.
During an investigation, the temperature of the beaker drops from 22°C to 15°C after two substances are combined. What does this suggest?
It suggests the reaction is endothermic because it absorbed/stored energy from the surroundings.
Why is it important to record the starting temperature before mixing substances in an investigation?
Because you need it to compare before and after and determine whether energy was released or stored.

Is ice melting endothermic or exothermic?
Endothermic. The ice is absorbing the heat from its surrounding.
 Endothermic or exothermic?
Endothermic or exothermic?
Exothermic. the heat energy is on the product side.
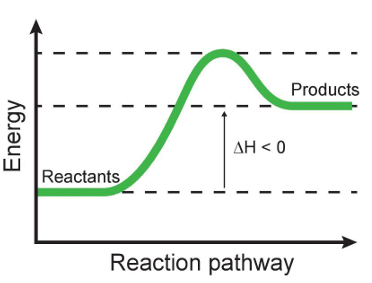 This graph represents an exothermic reaction.
This graph represents an exothermic reaction.
FIX IT: This graph shows an endothermic reaction.