Which of the following Group 5A elements is the most electronegative?
a. P
b. As
c. Bi
d. N
e. Sb
d. N
Of the bonds present in the molecule below, the triple C-N bond is predicted to have the ___ energy and the ___ length.

a. highest, longest
b. highest, shortest
c. lowest, longest
d. lowest, shortest
e. cannot compare w/ given info
b. highest, shortest
What is the formal charge on the sulfur atom in the sulfate ion?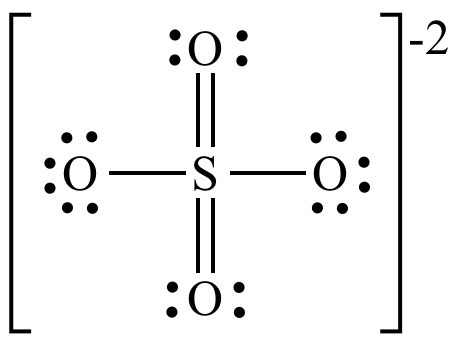
a. -2
b. -1
c. 0
d. +1
e. +2
c. 0
Which of the following answers has Period 3 oxide salts written in order of increasing lattice energy?
a. MgO < Na2O < Al2O3
b. MgO < Al2O3 < Na2O
c. Al2O3 < MgO < Na2O
d. Na2O < MgO < Al2O3
e. Al2O3 < Na2O < MgO
d. Na2O < MgO < Al2O3
Using the periodic table, determine the Lewis dot structure for phosphorus. How many paired and unpaired electrons are there?
a. 6 paired, 1 unpaired
b. 1 paired, 4 unpaired
c. 5 paired, 0 unpaired
d. 2 paired, 3 unpaired
e. 3 paired, 2 unpaired
d. 2 paired, 3 unpaired
Which of the following bonds is the most polar?
a. N-Br
b. C-Br
c. B-Br
d. O-Br
e. F-Br
e. F-Br
For the bromine ion, how many valence electrons are present and what is the overall charge?
a. 6 e-, +1
b. 7 e-, 0
c. 8 e-, -1
d. 4 e-, 0
e. 7 e-, -1
c. 8 e-, -1
Going from left to right, determine the hybridization for each non-H atom in the following molecule
:max_bytes(150000):strip_icc()/formaldehyde_LD-56a12a2c3df78cf772680353.png)
a. sp2, sp2
b. sp, sp2
c. sp3, sp2
d. sp3, sp3
e. sp2, sp
a. sp2, sp2
How many sigma and pi bonds are in the molecule CH2O?
a. 4 sigma, 0 pi
b. 3 sigma, 1 pi
c. 2 sigma, 2 pi
d. 1 sigma, 3 pi
e. 0 sigma, 4 pi
b. 3 sigma, 1 pi
What is the electron domain and molecular geometry for PO43-?
a. tetrahedral, tetrahedral
b. tetrahedral, trigonal pyramidal
c. trigonal bipyramidal, T-shaped
d. octahedral, square planar
e. trigonal bipyramidal, seesaw
a. tetrahedral, tetrahedral
Which set of orbitals forms the sigma bond between hydrogen and carbon in HCN?
a. s and s
b. s and sp
c. sp and sp
d. s and sp2
e. sp and sp2
b. s and sp
in an sp2-hybridized atom where there are 3 electron domains, __ s and __ p orbitals are combined to form __ sp2 orbital(s).
a. 2,2,3
b. 1,3,1
c. 1,3,4
d. 1,3,3
e. 1,2,3
e. 1,2,3
What is the correct Lewis structure for sulfur dioxide?
a. (refer to structure on board)
b.(refer to structure on board)
c.(refer to structure on board)
d.(refer to structure on board)
e.(refer to structure on board)
d.(refer to structure on board)
How many bonding and nonbonding domains are there for the center atom in CO32-?
a. 4 bonding, 0 nonbonding
b. 3 bonding, 0 nonbonding
c. 3 bonding, 1 nonbonding
d. 2 bonding, 2 nonbonding
e. 2 bonding, 1 nonbonding
b. 3 bonding, 0 nonbonding
Which of the following anions has a center atom that has more than an octet?
a. BO33-
b. SO42-
c. NO2-
d. CO32-
e. BrO-
b. SO42-
Which of the following molecules has an sp2-hybridized carbon?
I. HCN
II. CCl4
III. CH3Cl
IV. CH2O
a. I
b. I and IV
c. II and III
d. II and IV
e. IV only
e. IV only
How many resonance structures does PO43- (phosphate anion) have?
a. 0 (it has no resonance)
b. 1
c. 2
d. 3
e. 4
e. 4
Of the molecules listed below, which is non polar?
a. NH3
b. HF
c. SO2
d. F2
e. CHF3
d. F2