What is a Calorie a unit of?
A Calorie is a unit of energy
Name two Lab Safety Rules
no horseplay, long pants, pull hair back, ect
How many Joules are in 400 calories?
1680 Joules
Specific heat is the quantity of heat required to raise the temperature of Blank of a substance by one Blank
Specific heat is the quantity of heat required to raise the temperature of one gram of a substance by one Celsius degree
What are the 5 main food groups?
Dairy, Protein, Vegetables, Fruits and Grains
Why is it important to zero/ tare the scale before putting anything on it?
the weight of the object will be automatically subtracted from every reading until the balance is re-tared or zeroed. This helps you get the mass of the object you actually need.
How many Kilocalories (Calories) are in 5,678 Joules?
1.357 Kilocalories or Calories
In the equation: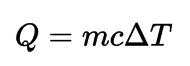 What does c stand for
What does c stand for
c = specific heat capacity
What is the 5/20 rule regarding nutrition?
If the %DV (Daily Value) is less than 5% there is a low amount of this nutrient, while if the %DV is greater than 20% there is a high amount of this nutrient.
In our burning food lab, what was one important lab technique that was discussed?
Keep thermometer off the bottom of the flask, Keep heat trapped inside, Proper weighing technique.
If a bag of chips has 200 Calories per serving in it and a serving is 5 chips, how many calories are in 20 chips?
(Note: Calories are different than calories)
800,000 calories
If 50 grams of water (m) were heated by 2 degrees Celsius (delta T), what is the energy in calories produced or used? Note: c= 1cal/g Celsius
100 calories
How many Joules are released if the mass of water is 50g and the change in temp was 30 degrees Celsius?
6,300 Joules
Name two or more ways protein helps your body
building block for bones/ muscles. supports the immune system. reduces the risk of heart disease
Name one source of error from the experiment
heat lost to the can and to the air. Some of the heat was transferred to the can to warm it up, and some may have been transferred to the air between the food and the can.
The thermometer may have measured the heat of the can and not the water
The sample could not have been thoroughly burned
If one cup of rice has 150 Calories in it, and you eat 4 cups of this rice how many Joules of energy are consumed from this food?
2,520,000 Joules
What is the mass of water if the change in temp is 4 degrees Celsius (delta T) and the energy is 280 calories (Q)? Note: c= 1cal/g Celsius
70 grams of water
How many Joules are released if the mass of water is 20g and the final temp and initial temp were respectively 30 degrees celsius and 20 degrees celsius
840 Joules
Name three Vitamins/ Nutrients that are a part of Dairy.
calcium, phosphorus, vitamin A, Vitamin D, riboflavin, vitamin B12, protein, potassium zinc, Choline, magnesium, or selenium
Name one way we could have improved the experiment.
better equipment, more trails, including more control, have better measurement techniques ect.
What is the difference in the energy in Joules between these two foods for one serving?
Food 1. Food 2
Calories: 342 Calories 453
1,436,400 joules - 1,902,600 joules
-466,200 Joules
What is the final temperature if Q=250 calories, m = 50 grams, and the initial temperature is 25 degrees Celsius? Note: c= 1cal/g Celsius
30 degrees Celcius
What is the energy in Joules in this food?
Food info:
mass = 0.0005kg
T final = 40 degrees celsius
T initial = 20 degrees celsius
420 Joules